
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Brain cancer encompasses malignant tumors that develop in the brain or other parts of the central nervous system, often leading to severe neurological impairment as they grow and spread. According to recent estimates, 187,162 people were living with brain and other nervous system cancers in the United States in 2022, highlighting its substantial disease burden. Treatment approaches include surgery, radiation therapy, chemotherapy, targeted therapy, and emerging immunotherapies designed to control tumor progression and improve survival. The brain cancer pipeline analysis by Expert Market Research reveals ongoing advancements in molecular profiling and innovative therapeutic platforms for brain cancer. There is an emphasis on offering new opportunities for more precise and effective treatment strategies in the coming years.
Major companies involved in the brain cancer pipeline analysis include Pfizer, and Jazz Pharmaceuticals, and others.
Leading drugs currently in the pipeline include Levetiracetam, 5-ALA (Gliolan), and others.
Brain cancer growth is primarily driven by rising diagnostic capabilities, increasing exposure to genetic and environmental risk factors, and improved detection of central nervous system tumors.
The Brain Cancer Pipeline Analysis Report by Expert Market Research gives comprehensive insights into brain cancer therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for Brain Cancer. The brain cancer report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The brain cancer pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with brain cancer treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to brain cancer.
Read more about this report - Request a Free Sample
Brain cancer encompasses a diverse group of malignant tumors that arise within the brain or central nervous system, often leading to significant neurological impairment as they grow and infiltrate vital structures. These tumors develop due to genetic mutations, environmental influences, and disruptions in normal cellular regulation that drive uncontrolled proliferation and aggressive progression. High-grade gliomas, pediatric brain tumors, and diffuse midline gliomas remain among the most challenging to treat, highlighting the urgent need for more effective and targeted therapeutic strategies.
Brain cancer treatment includes surgery, radiation therapy, chemotherapy, targeted agents, and emerging immunotherapies designed to slow tumor growth, enhance survival, and manage debilitating symptoms. Several FDA-approved therapies are shaping the current treatment landscape, including temozolomide for newly diagnosed glioblastoma, bevacizumab for recurrent glioblastoma, and the 2024 approval of vorasidenib for IDH-mutant low-grade gliomas, as well as the 2025 accelerated approval of dordaviprone for H3 K27M-mutant diffuse midline glioma. With additional molecularly targeted agents and immune-modulating therapies advancing through clinical development, the brain cancer pipeline is rapidly evolving and expected to transform future standards of care.
Brain cancer represents a significant and growing health concern in the United States, with an estimated 187,162 individuals living with brain and other nervous system tumors in 2022. This prevalence reflects both the increasing incidence of certain tumor types and improvements in diagnostic imaging that allow earlier and more accurate detection. Despite advancements in treatment, many brain cancers remain aggressive and difficult to manage, contributing to long-term neurological challenges and reduced quality of life for affected individuals. The substantial number of patients living with these tumors underscores the ongoing need for innovative therapies, improved care strategies, and continued research efforts to better address the burden of brain cancer.
This section of the report covers the analysis of brain cancer drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
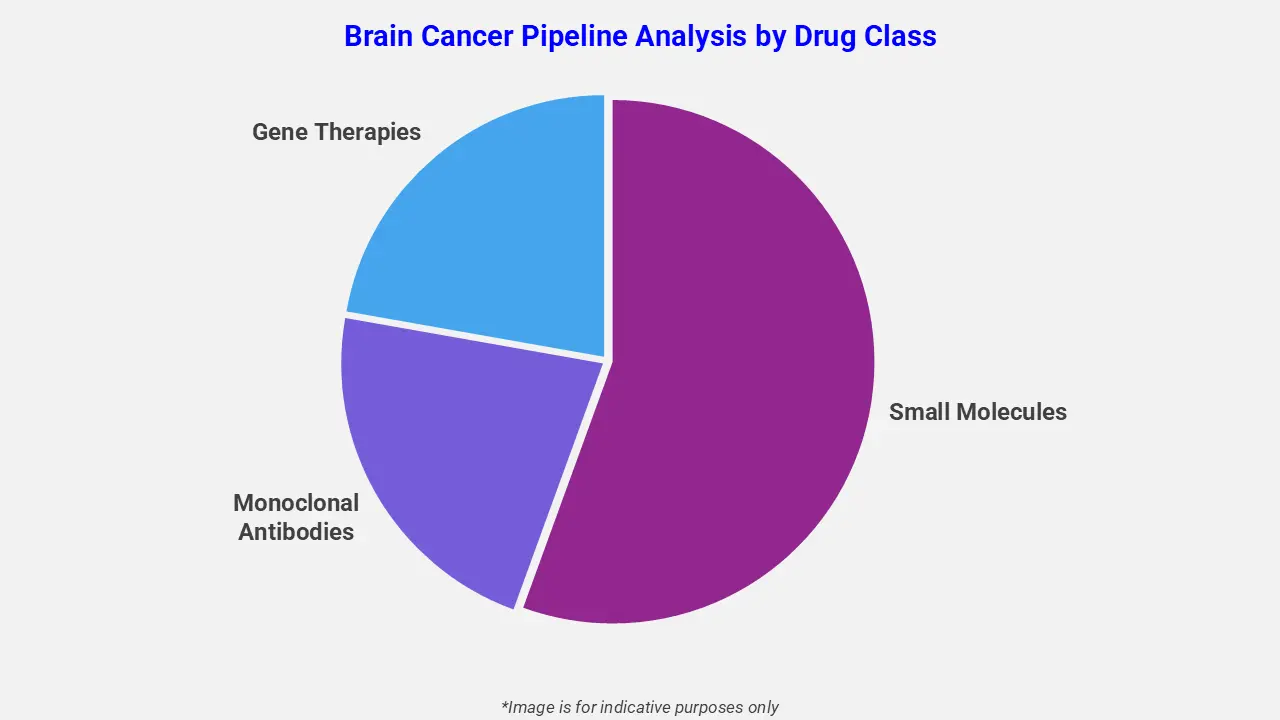
By Drug Class
The brain cancer pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with 53%, covers a major share of the total brain cancer clinical trials. It is followed by phase I at 38% and other phases. The strong representation in Phase I and II highlights robust clinical development, signaling potential approvals, increased treatment options, and growth opportunities in the therapeutic landscape.
The drug molecule categories covered under the brain cancer pipeline analysis include small molecules, monoclonal antibodies, and gene therapies. The brain cancer report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for Brain Cancer. For example, Niraparib, a small-molecule PARP inhibitor, is being evaluated in a Phase III trial comparing its efficacy with temozolomide in adult patients with newly diagnosed, MGMT-unmethylated glioblastoma. As a small-molecule targeted therapy, Niraparib aims to inhibit DNA repair pathways in tumor cells, enhancing treatment response and offering a promising addition to the brain cancer pipeline.
The EMR report for the brain cancer pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed brain cancer therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in brain cancer clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for brain cancer. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of brain cancer drug candidates.
Levetiracetam is an antiepileptic drug being investigated in a Phase III clinical study sponsored by Tata Memorial Centre to evaluate its optimal duration of use in patients with brain tumors who experience seizures. This study aims to determine whether short-term or long-term administration of levetiracetam provides better seizure control and overall clinical outcomes. As a widely used anticonvulsant, levetiracetam modulates synaptic neurotransmission to reduce neuronal hyperexcitability, helping manage tumor-associated seizures. Administered orally or intravenously, it is being assessed for its efficacy, safety, and impact on quality of life in individuals with brain tumors requiring seizure management.
5-ALA (Gliolan) is a fluorescent-guided surgical imaging agent being evaluated in a Phase II clinical study sponsored by University College London in collaboration with the University of Nottingham for pediatric patients with high-grade brain tumors. This study aims to determine whether 5-ALA can enhance tumor visualization during surgery, enabling more precise resection and improving clinical outcomes. After administration, 5-ALA is metabolized into a fluorescent compound that accumulates in malignant cells, allowing surgeons to distinguish tumor tissue under specialized lighting. Administered orally, it is being assessed for its safety, efficacy, and potential to optimize surgical accuracy in pediatric neuro-oncology.
NEO100 is an investigational therapeutic agent developed by Neonc Technologies, Inc., currently set to begin evaluation in a Phase I clinical study for pediatric patients with central nervous system tumors, including diffuse midline glioma, high-grade glioma, and other rare brain and spinal cord malignancies. This study aims to assess the safety, optimal dosing, and delivery of NEO100, which is derived from a purified form of perillyl alcohol designed to penetrate the blood-brain barrier more effectively. Administered intranasally, NEO100 is being evaluated for its potential anti-tumor activity, safety profile, pharmacokinetics, and feasibility as a novel treatment option for pediatric brain tumors with limited therapeutic alternatives.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Brain Cancer Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for Brain Cancer. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into brain cancer collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share