
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Gaucher disease type 1 (GD1) is a non‑neuronopathic lysosomal storage disorder caused by deficient glucocerebrosidase, leading to fat accumulation in organs like the liver, spleen, and bone marrow. According to Daniela Anahí Méndez‑Cobián et al. (2024), its incidence is about 1 case per 40,000–60,000 births in the general population. Existing therapies include enzyme replacement therapy (ERT) (e.g., imiglucerase, velaglucerase) and oral substrate reduction therapy (SRT) (e.g., miglustat). According to the Gaucher disease type‑1 pipeline analysis by Expert Market Research, there is a growing pipeline of novel agents, including next‑generation ERTs, gene therapies, and small molecules, reflecting increasing R&D focus. This robust pipeline, coupled with rising disease awareness and supportive regulatory frameworks, is expected to drive significant market growth in the coming years.
Major companies involved in the Gaucher disease type 1 pipeline analysis include Spur Therapeutics, Prevail Therapeutics, and others.
Leading drugs currently in the pipeline include FLT201, LY3884961, LY-M001, and others.
The pipeline is poised for growth driven by advancements in enzyme replacement therapies, increasing development of substrate reduction treatments, and expanding clinical trials targeting improved efficacy and patient outcomes.
The Gaucher Disease Type 1 Pipeline Analysis Report by Expert Market Research gives comprehensive insights into Gaucher disease type 1 therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for Gaucher disease type 1. The Gaucher disease type 1 report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The Gaucher disease type 1 pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with Gaucher disease type 1 treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to Gaucher disease type 1.
Read more about this report - Request a Free Sample
Gaucher disease type 1 is a hereditary lysosomal storage disorder caused by a deficiency of the enzyme glucocerebrosidase. This deficiency leads to the accumulation of glucocerebroside in macrophages, resulting in enlarged liver and spleen, anemia, bone pain, and skeletal abnormalities. The condition primarily affects adolescents and adults, with progressive multi-organ involvement that can become life-threatening if untreated.
Gaucher disease type 1 treatments focus on enzyme replacement therapy using recombinant human glucocerebrosidase, which significantly improves organ function, alleviates hematological symptoms, and enhances patients' quality of life. In May 2025, Velaglucerase beta for injection (CAN103) received NMPA approval. Developed through Phase I/II trials led by PUMCH, this innovative enzyme replacement therapy provides long-term treatment for adolescents aged 12 years and older and adults, demonstrating significant efficacy, favorable safety, and sustained clinical benefits.
The data on Gaucher disease type 1 highlight its epidemiological significance and impact on drug development. According to Daniela Anahí Méndez-Cobián et al., 2024, Gaucher disease is the most common lysosomal storage disorder, with an incidence of 1 case per 40,000–60,000 births globally and 0.125% of births among the Ashkenazi Jewish population. According to Gaucher disease type 1 epidemiology forecast by Expert Market Research, over 90% of patients in North America and Europe have type 1, presenting primarily with splenomegaly, hepatomegaly, and blood and bone abnormalities. As per Aya Narita et al., 2024, Japan has a prevalence of 1 in 330,000, affecting approximately 211 patients. These data underscore the need for continued drug development.
This section of the report covers the analysis of Gaucher disease type 1 drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
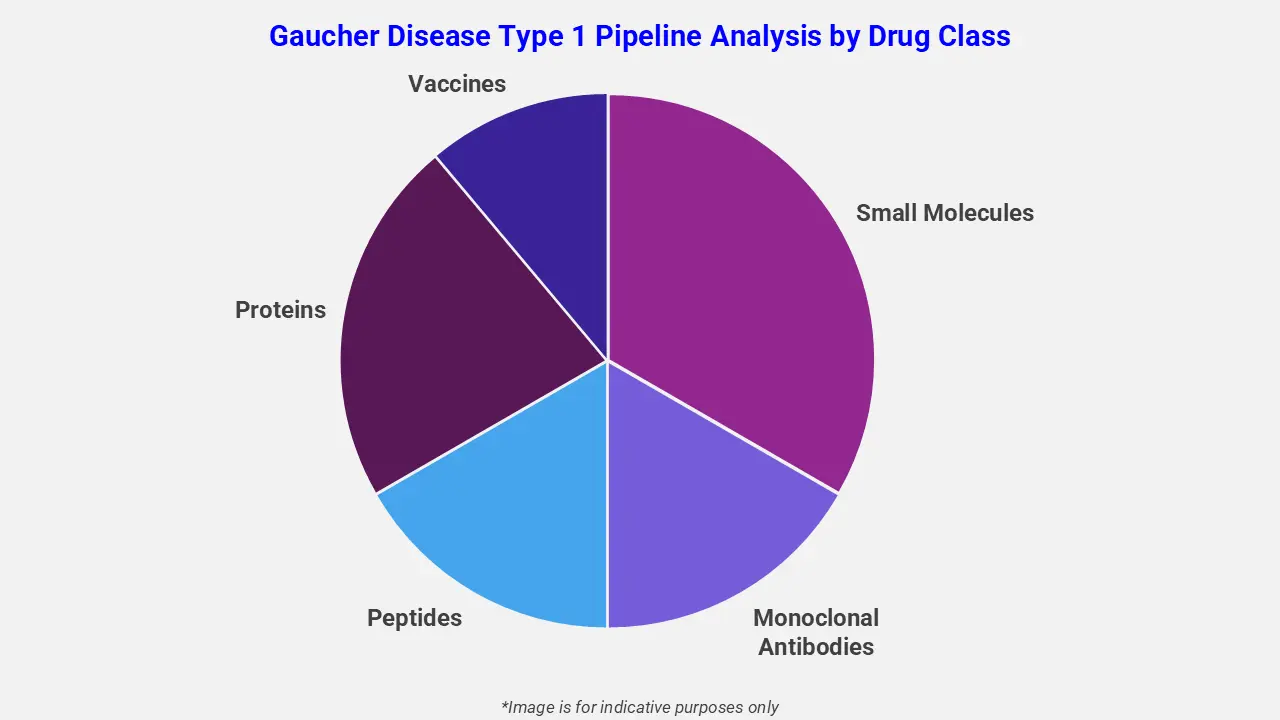
By Drug Class
The Gaucher disease type 1 pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration.
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II with 50%, covers a major share of the total Gaucher disease type 1 clinical trials, reflecting strong ongoing clinical development and potential near-future therapeutic advancements. Phase I contributes 33%, highlighting promising early-stage candidates. Phase III represents 11%, indicating progress toward late-stage approval. These active phases collectively support growth and expansion in the Gaucher disease type 1 treatment landscape.
The drug molecule categories covered under the Gaucher disease type 1 pipeline analysis include small molecules, monoclonal antibodies, peptides, proteins, and vaccines. The Gaucher disease type 1 report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for Gaucher disease type 1. Enzyme replacement therapies and substrate reduction therapies are the main drug classes under investigation for Gaucher Disease Type 1. For instance, eliglustat, an oral substrate reduction therapy, is being studied alone or in combination with imiglucerase, an intravenous enzyme replacement therapy, to evaluate safety and efficacy in pediatric patients. These approaches aim to reduce fat accumulation in organs and improve hematological and bone-related symptoms.
The EMR report for the Gaucher disease type 1 pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed Gaucher disease type 1 therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in Gaucher disease type 1 clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for Gaucher disease type 1. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of Gaucher disease type 1 drug candidates.
FLT201 is a Phase 3 gene therapy sponsored by Spur Therapeutics, specifically designed for Gaucher disease type 1. This therapy uses an adeno-associated virus (AAV) vector, AAVS3, carrying an engineered GBA1 transgene that encodes the unique glucocerebrosidase variant GCase85. GCase85 features an engineered disulfide, increasing its active half-life by over six-fold in human serum and over twenty-one-fold in lysosomal pH, while retaining the catalytic properties of wild-type GCase. The study aims to confirm FLT201’s efficacy and safety after discontinuation of enzyme replacement or substrate reduction therapy. Administration is via gene therapy infusion, with an estimated completion in 2032.
LY3884961 is a gene therapy sponsored by Prevail Therapeutics, currently being evaluated in a Phase 1/2 clinical trial (PROCEED) for patients with peripheral manifestations of Type 1 Gaucher disease. This innovative therapy uses an adeno-associated viral vector serotype 9 to deliver a codon-optimized human GBA gene, aiming to restore the production of the deficient enzyme glucocerebrosidase. The study is examining the safety, tolerability, immunogenicity, biomarkers, and efficacy of a single intravenous dose of LY3884961. The trial is enrolling approximately 15 patients, with an estimated completion set for October 2030, and includes long-term follow-up over five years.
LY-M001 injection is an innovative rAAV8 vector gene therapy being developed by Lingyi Biotech Co., Ltd. for adult patients with Gaucher disease type 1. This therapy is designed to transduce the liver after a single intravenous administration and enable long-term expression of the glucocerebrosidase (GCase) protein, addressing the underlying enzyme deficiency caused by GBA1 mutations. The Phase I/II study is evaluating the safety, tolerability, efficacy, immunogenicity, and pharmacokinetics of LY-M001 through a multicenter, open-label, single-arm, dose-escalation design. The trial, started in July 2024, is expected to reach primary completion by July 2027 and overall completion by July 2031, enrolling approximately 18 participants.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Gaucher Disease Type 1 Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for Gaucher disease type 1. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into Gaucher disease type 1 collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share