
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Neuronal ceroid lipofuscinoses (NCL), commonly known as Batten disease, are a group of inherited neurodegenerative disorders characterized by progressive loss of motor and cognitive functions. According to Aparna Bose et al., 2024, Batten disease is the most common neurodegenerative disorder in childhood, with a global prevalence of roughly 1 in 100,000 live births. The NCL drug pipeline includes enzyme replacement, gene therapies, small molecules, and antisense oligonucleotides targeting specific genetic mutations. According to the neuronal ceroid lipofuscinoses pipeline analysis by Expert Market Research, growing research focus, regulatory advancements, and increased funding are expected to drive significant therapeutic developments in the coming years.
Major companies involved in the neuronal ceroid lipofuscinoses pipeline analysis include Polaryx Therapeutics, Inc., Tern Therapeutics, LLC, and others.
Leading drugs currently in the pipeline include PLX-200, TTX-381, AAV9/CLN7, and others.
The pipeline shows accelerated growth driven by emerging gene therapies, increased focus on rare pediatric indications, and expanding clinical trials targeting multiple NCL subtypes, enhancing treatment options and disease management.
The Neuronal Ceroid Lipofuscinoses Pipeline Analysis Report by Expert Market Research gives comprehensive insights into neuronal ceroid lipofuscinoses therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for neuronal ceroid lipofuscinoses. The neuronal ceroid lipofuscinoses report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The neuronal ceroid lipofuscinoses pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with neuronal ceroid lipofuscinoses treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to neuronal ceroid lipofuscinoses.
Read more about this report - Request a Free Sample
Neuronal ceroid lipofuscinoses (NCLs) are rare, inherited lysosomal storage disorders causing progressive neurodegeneration. Mutations in genes such as TPP1 lead to deficient enzyme activity, resulting in lipofuscin accumulation in neurons. This buildup disrupts brain and retinal functions, causing cognitive decline, motor deterioration, seizures, speech and language delays, and vision loss. The symptoms typically appear in early childhood, with disease progression leading to severe disability and reduced life expectancy.
Neuronal ceroid lipofuscinoses treatments primarily involve enzyme replacement and supportive care. Brineura® (cerliponase alfa) slows loss of ambulation, reduces neurodegeneration, and preserves motor function in children with CLN2 disease. In July 2024, BioMarin demonstrated that intraventricular Brineura® administration in children under three years effectively delays disease onset and motor decline, representing a significant advancement in early CLN2 intervention.
The pipeline is expanding, driven by the need to address its diverse genetic and phenotypic variations. According to Aparna Bose et al., 2024, Batten disease, the most common childhood neurodegenerative disorder, has a global prevalence of approximately 1 in 100,000 live births, with higher incidences in Scandinavian countries like Finland. Juvenile NCL (CLN3) is the most frequent form worldwide, whereas CLN1 (Santavuori-Haltia disease) is more common in Finland, with a 1 in 20,000 incidence. As per the Cleveland Clinic, about 3 of every 100,000 babies in the United States are affected. These epidemiological insights guide targeted drug development and early diagnostic strategies.
This section of the report covers the analysis of neuronal ceroid lipofuscinoses drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
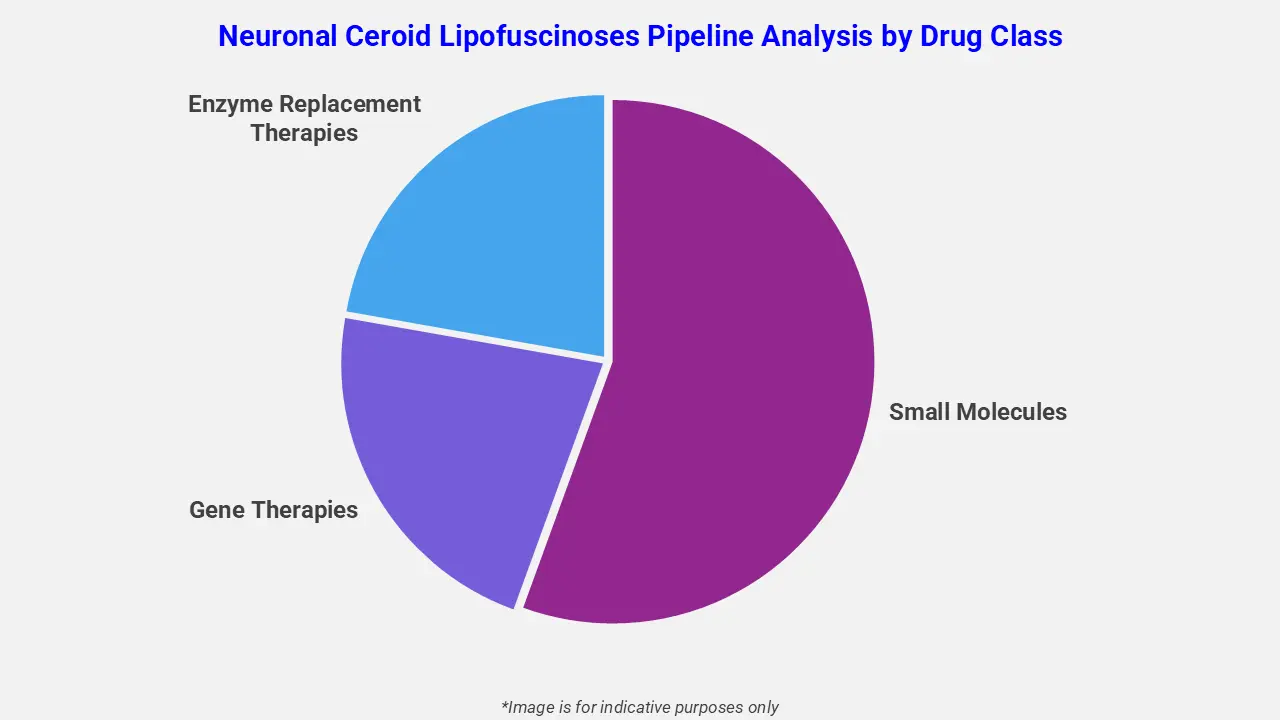
By Drug Class
The neuronal ceroid lipofuscinoses pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase I covers a major share of the total neuronal ceroid lipofuscinoses clinical trials. Phase I leads with 55%, reflecting strong early clinical activity and robust investigational focus. Phase II represents 36%, indicating significant progression of promising candidates. Phase III contributes 10%, showcasing advanced-stage therapies nearing potential approval. This distribution positively underscores innovation and pipeline depth, supporting future market growth and treatment availability.
The drug molecule categories covered under the neuronal ceroid lipofuscinoses pipeline analysis include small molecules, gene therapies, and enzyme replacement therapies. The neuronal ceroid lipofuscinoses report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for neuronal ceroid lipofuscinoses. Gene-targeted therapies in the pipeline are advancing to address the underlying genetic defects in patients. For instance, Batten-1 (miglustat) is under investigation for CLN3 disease, reducing glycosphingolipid accumulation and neuroinflammation. Moreover, antisense oligonucleotides and enzyme replacement approaches are being evaluated to slow neuronal degeneration and preserve cognitive and motor functions in affected individuals.
The EMR report for the neuronal ceroid lipofuscinoses pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides detailed neuronal ceroid lipofuscinoses therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in neuronal ceroid lipofuscinoses clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for neuronal ceroid lipofuscinoses. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of neuronal ceroid lipofuscinoses drug candidates.
PLX-200 is an oral small-molecule therapy being developed by Polaryx Therapeutics, Inc. for juvenile neuronal ceroid lipofuscinosis (CLN3). The drug is administered as a 15 mg/mL oral solution, taken twice daily via syringe approximately 30 minutes before meals. PLX-200 works as a PPARα agonist, enhancing lysosome biogenesis through TFEB upregulation, reducing neuroinflammation, and promoting neuronal survival. This Phase 3, double-blind, placebo-controlled study examines the safety, tolerability, pharmacokinetics, and efficacy of escalating weight-based doses over a 60-week maintenance period, followed by a 36-week open-label extension. The study is set to be completed by March 31, 2026, with an estimated enrollment of 39 participants.
TTX-381 is an investigational gene therapy being developed by Tern Therapeutics, LLC for the treatment of ocular manifestations in neuronal ceroid lipofuscinosis type 2 (CLN2) disease. This first-in-human, open-label, single ascending dose study examines the safety and tolerability of TTX-381, which delivers a functional copy of the TPP1 gene directly to the retina via ocular administration. The therapy is designed to restore continuous production of the TPP1 enzyme, preventing harmful buildup in retinal cells and potentially preserving vision. The study enrolls 16 participants, with estimated primary completion by July 2026 and overall study completion in July 2031.
AAV9/CLN7 is a gene therapy agent being developed for the treatment of CLN7 Batten disease, sponsored by Benjamin Greenberg at the University of Texas Southwestern Medical Center. This Phase 1, open-label study is administering a single intrathecal injection into the lumbar spinal cord of pediatric patients, with two dosing cohorts: a low dose of 5×10¹⁴ vg and a high dose of 1×10¹⁵ vg. The therapy uses an AAV9 vector to deliver a fully functional MFSD8 gene under a synthetic promoter, aiming for stable, long-term expression in non-dividing cells. The study is examining safety, motor and cognitive function, visual impairment, and brain activity, with estimated completion in February 2029.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Neuronal Ceroid Lipofuscinoses Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for neuronal ceroid lipofuscinoses. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into neuronal ceroid lipofuscinoses collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share