
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Retinal edema is characterized by fluid accumulation within the retinal layers, resulting in swelling and progressive vision impairment. According to Özlem Candan et al., 2025, macular edema affects slightly more than half of individuals with retinal vein occlusion, accounting for approximately 52.5% of cases, with a 95% confidence interval ranging from 47.48% to 57.56%. According to the retinal edema pipeline analysis by Expert Market Research, the pipeline is advancing with multiple candidates across early- and late-stage development. Current therapies include anti-VEGF agents, corticosteroids, and sustained-release delivery systems. The increasing emphasis on targeted biologics, innovative drug delivery technologies, and the rising retinal disorder incidence is expected to support robust pipeline growth in the coming years.
Major companies involved in the retinal edema pipeline analysis include Kodiak Sciences Inc., Kiora Pharmaceuticals, Inc., and others.
Leading drugs currently in the pipeline include KSI-101, KIO-104, ABBV-RGX-314, and others.
The robust pipeline growth is driven by sustained-release anti-VEGF formulations, novel corticosteroid implants, and gene-based therapies targeting vascular permeability, alongside expanded clinical trials addressing diabetic macular edema and post-surgical complications.
The Retinal Edema Pipeline Analysis Report by Expert Market Research gives comprehensive insights into retinal edema therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for retinal edema. The retinal edema report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The retinal edema pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with retinal edema treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to retinal edema.
Read more about this report - Request a Free Sample
Retinal edema is a condition characterized by fluid accumulation in the retina, often caused by retinal vein occlusion, diabetic retinopathy, or inflammation. The excess fluid leads to swelling, impaired vision, and distortion of the retinal structure, potentially resulting in vision loss if untreated. It occurs when the blood-retinal barriers are compromised, allowing leakage of fluids and proteins into the retinal tissue.
Retinal edema treatments include anti-vascular endothelial growth factor injections, corticosteroid therapies, and laser photocoagulation. These interventions aim to reduce retinal swelling, improve visual acuity, and prevent disease progression, with dosing schedules tailored to individual patient response and severity. In November 2025, Regeneron’s EYLEA HD 8 mg injection demonstrated strong efficacy in the retinal edema pipeline landscape, showing non-inferior visual acuity gains and reduced injection frequency in patients with retinal vein occlusion, as confirmed in the phase 3 QUASAR trial.
The pipeline shows significant growth potential due to the rising burden of diabetic eye disease. According to Özlem Candan et al., 2025, macular edema affects 52.52% of patients with retinal vein occlusion. According to Genentech, diabetic macular edema impacts approximately 29 million people worldwide. According to Yuan, Yuan et al., 2024, diabetic retinopathy accounts for 89.53% of research grants focused on diabetic eye disease, while 58.19% of clinical trials target diabetic macular edema. Despite these efforts, few new drugs have been approved, highlighting an urgent need for innovative therapeutics in retinal edema management.
This section of the report covers the analysis of retinal edema drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
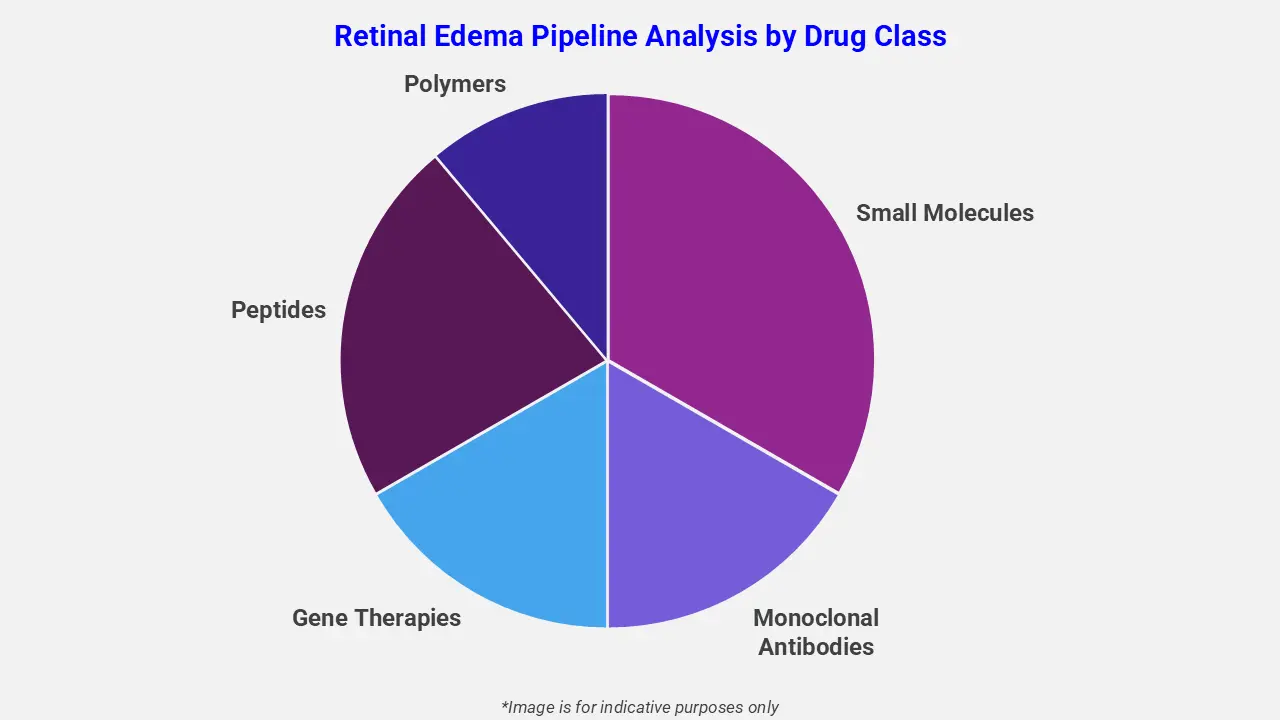
By Drug Class
The retinal edema pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with 31%, covers a major share of the total retinal edema clinical trials, highlighting significant mid-stage development activity that can drive innovation and market expansion. Phase III contributes 26%, supporting late-stage advancements and potential approvals. Phase I holds 21%, early phase I accounts for 10%, and phase IV represents 12%, reflecting ongoing post-marketing studies that enhance treatment optimization and patient outcomes.
The drug molecule categories covered under the retinal edema pipeline analysis include small molecules, monoclonal antibodies, gene therapies, peptides, and polymers. The retinal edema report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for retinal edema. Anti-vascular endothelial growth factor (VEGF) therapies are leading the retinal edema drug pipeline, targeting abnormal blood vessel growth and leakage in the retina. For instance, Roche’s Vabysmo® (faricimab) is a bispecific antibody inhibiting both angiopoietin-2 and VEGF-A pathways, showing sustained vision improvements and retinal drying in neovascular age-related macular degeneration and polypoidal choroidal vasculopathy.
The EMR report for the retinal edema pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed retinal edema therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in retinal edema clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for retinal edema. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of retinal edema drug candidates.
KSI-101 (Tabirafusp Alfa) is a novel, high-strength (100 mg/mL) bispecific protein that targets interleukin-6 (IL-6) and vascular endothelial growth factor (VEGF), key mediators of inflammation and vascular leakage in retinal disease. Sponsored by Kodiak Sciences Inc, this randomized, double-masked, sham-controlled Phase 3 PINNACLE study is evaluating the efficacy and safety of intravitreal KSI-101 in patients with macular edema secondary to inflammation (MESI). The drug is being administered via intravitreal injection to reduce retinal fluid and improve visual outcomes. Primary completion is estimated in December 2026, with overall study completion anticipated in July 2027.
KIO-104 is a potent, locally delivered small molecule designed to treat inflammatory retinal diseases, including macular edema. Administered via intravitreal injection, KIO-104 works by inhibiting the mitochondrial enzyme DHODH, reducing T-cell replication, and suppressing pro-inflammatory cytokines that drive retinal damage. The Phase 2 KLARITY-1 study, sponsored by Kiora Pharmaceuticals, Inc., is assessing the safety, tolerability, and efficacy of multiple KIO-104 doses in the study eye, with dosing intervals every 2 or 4 weeks. Enrollment includes 28 participants, and the study is expected to be completed by December 2026.
RGX-314 is a novel gene therapy being developed by AbbVie for the treatment of Diabetic Retinopathy (DR) with and without center-involved Diabetic Macular Edema (CI-DME). Administered via a single suprachoroidal space (SCS) injection, RGX-314 uses the NAV® AAV8 vector to deliver an antibody fragment that inhibits vascular endothelial growth factor (VEGF), preventing abnormal blood vessel growth and fluid accumulation in the retina. This Phase II, randomized, dose-escalation study is examining the drug’s safety, tolerability, and efficacy in approximately 139 participants. The study is expected to be completed in December 2026, with participants receiving either RGX-314 or comparator treatments.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Retinal Edema Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for retinal edema. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into retinal edema collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share