
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Soft tissue sarcoma is a rare malignancy that accounts for less than 1% of all adult cancers and represents a notable proportion of pediatric solid tumors. In the United States, thousands of new cases are diagnosed annually, with rates varying by age and histologic subtype. Data show incidence increases with age, and survival outcomes differ widely due to tumor heterogeneity and treatment challenges. The soft tissue sarcoma pipeline analysis by Expert Market Research highlights emerging targeted and immunotherapy approaches in development, reflecting efforts to improve prognosis and address unmet needs for this uncommon but impactful cancer.
Major companies involved in the soft tissue sarcoma pipeline analysis include Jazz Pharmaceuticals, Polaris Group, and others.
Leading drugs currently in the pipeline include NKTR-214, TK216, and others.
The soft tissue sarcoma pipeline is driven by advancements in precision oncology, including targeted therapies and novel immunotherapies. Increasing identification of actionable genetic mutations and development of biomarker-driven clinical trials are fueling growth, enabling personalized treatment approaches, improving efficacy, and expanding options for patients with rare and heterogeneous sarcoma subtypes.
The soft tissue sarcoma Pipeline Analysis Report by Expert Market Research gives comprehensive insights into soft tissue sarcoma therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for soft tissue sarcoma. The soft tissue sarcoma report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The soft tissue sarcoma pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with soft tissue sarcoma treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to soft tissue sarcoma.
Read more about this report - Request a Free Sample
The soft tissue sarcoma (STS) pipeline is evolving with innovative therapies targeting rare and aggressive subtypes. Traditionally, treatment has relied on surgery, radiotherapy, and chemotherapy, with limited efficacy in advanced cases.
Soft tissue sarcoma treatment achieved a significant advancement in August 2024, when the U.S. FDA granted accelerated approval to Tecelra (afamitresgene autoleucel), the first T-cell receptor (TCR) immunotherapy for adults with unresectable or metastatic synovial sarcoma expressing MAGE A4. Tecelra utilizes engineered autologous T cells to selectively target MAGE A4, representing a novel therapeutic mechanism and offering new hope for patients with refractory disease. This approval underscores the growing emphasis on precision immunotherapy within the soft tissue sarcoma development pipeline.
Soft tissue sarcomas (STS) are rare cancers arising from fat, muscle, and connective tissues, accounting for less than 1% of all adult solid tumors but about 7-8% of childhood cancers. STS incidence has increased globally over recent decades, though age‑standardized rates have slightly declined, and the burden remains higher in older adults and males. In the United States, an estimated 13,520 new STS cases are expected in 2025, with 5,420 deaths projected. These tumors’ heterogeneity and rarity underscore the importance of continued surveillance and research into tailored therapies and early detection.
This section of the report covers the analysis of soft tissue sarcoma drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
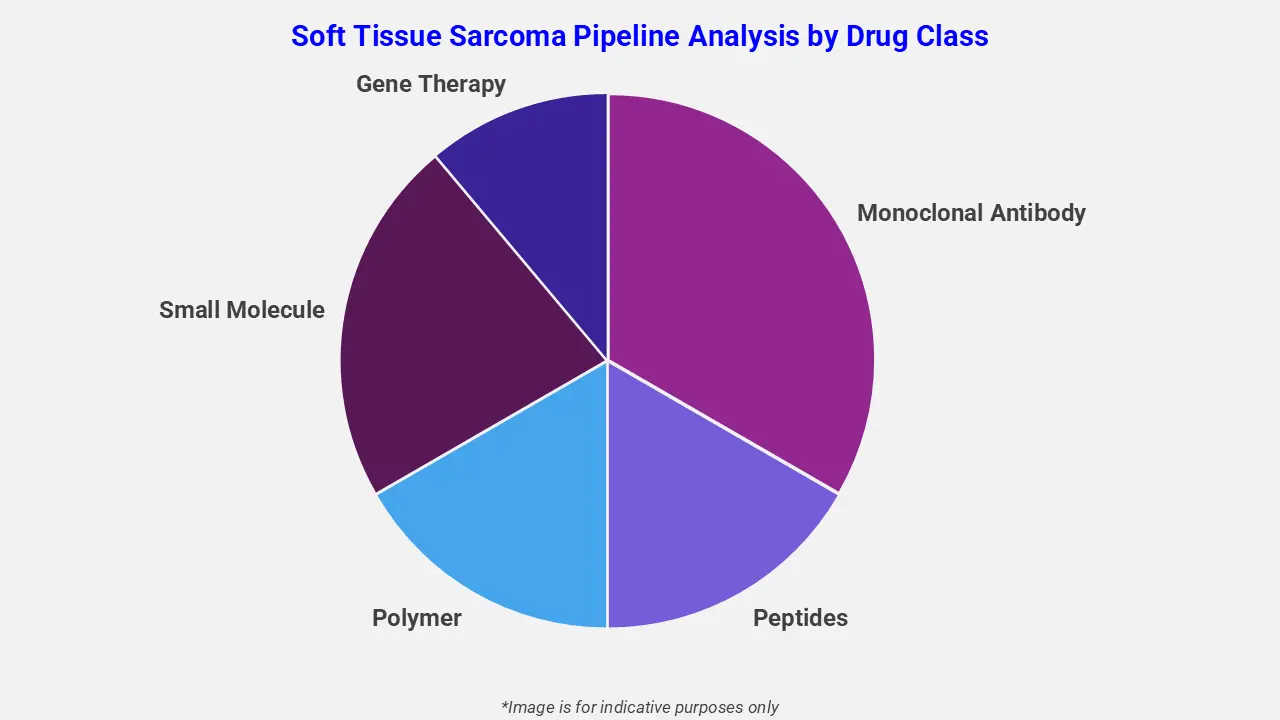
By Drug Class
The soft tissue sarcoma pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total soft tissue sarcoma clinical trials. The report covers phase I (36%), phase II (53%), and phase III (7%). Overall, this phase distribution highlights the ongoing clinical trial activity in the soft tissue sarcoma landscape.
The drug molecule categories covered under the soft tissue sarcoma pipeline analysis include monoclonal antibodies, peptides, polymers, gene therapies, and small molecules. The soft tissue sarcoma report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for soft tissue sarcoma. In August 2023, the U.S. Food and Drug Administration granted Orphan Drug Designation to tigilanol tiglate for the treatment of soft tissue sarcoma. This designation aims to support the development of tigilanol tiglate as a potential therapy for this rare cancer by providing incentives such as market exclusivity, tax credits, and clinical development support, reflecting ongoing efforts to expand treatment options for patients with limited therapeutic alternatives.
The EMR report for the soft tissue pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed soft tissue therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in soft tissue sarcoma clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for soft tissue sarcoma. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of soft tissue sarcoma drug candidates.
ION363 is an experimental antisense oligonucleotide (ASO) designed to target specific immune pathways implicated in neuroinflammatory and autoimmune conditions. By selectively binding to its target receptor, it modulates aberrant immune responses, potentially reducing tissue damage. Ionis Pharmaceuticals, known for pioneering antisense and RNA-targeted therapies, is conducting preclinical and early clinical studies to evaluate ION363’s safety, pharmacokinetics, and efficacy, aiming to offer a novel treatment option for patients with conditions resistant to conventional immunomodulatory therapies.
MK-1084 is a small-molecule inhibitor targeting a key enzymatic pathway involved in chronic inflammatory and fibrotic diseases. It works by selectively suppressing overactive cellular signaling, potentially mitigating tissue inflammation and fibrosis. Merck & Co., a global leader in pharmaceutical innovation, is spearheading clinical trials assessing MK-1084’s efficacy, dosage optimization, and long-term safety, with the goal of introducing a targeted therapy addressing unmet needs in patients with progressive inflammatory disorders.
PM14 is a peptide-based therapeutic candidate developed to modulate specific cellular pathways implicated in metabolic and cardiovascular diseases. By enhancing or inhibiting protein interactions, PM14 aims to restore normal cellular functions and prevent disease progression. Pharmaron, a contract research and development organization, is overseeing PM14’s preclinical studies, focusing on pharmacodynamics, safety profiling, and delivery mechanisms, supporting the development of a next-generation therapy with the potential to improve patient outcomes in metabolic syndrome and related chronic conditions.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Soft Tissue Sarcoma Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for soft tissue sarcoma. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into soft tissue sarcoma collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.