
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Thymic carcinoma is a rare and aggressive malignancy arising from epithelial cells of the thymus gland located in the anterior mediastinum. According to Tiemo Sven Gerber et al., 2024, epidemiological studies indicate that thymic carcinomas occur in approximately 0.42-0.48 cases per million individuals annually, highlighting their extremely low prevalence. According to the thymic carcinoma pipeline analysis by Expert Market Research, the therapeutic landscape includes surgery, chemotherapy, radiotherapy, immune checkpoint inhibitors, and emerging targeted therapies. The growing research on molecular profiling, biomarkers, and immunotherapy combinations is accelerating pipeline development.
Major companies involved in the thymic carcinoma pipeline analysis include Chongqing Precision Biotech Co., Ltd., Novartis Pharmaceuticals, and others.
Leading drugs currently in the pipeline include PT-112, M7824, CD70-targeted CAR-T cells, and others.
The increasing development of targeted therapies and immune checkpoint inhibitors, along with rising clinical trials for rare thoracic malignancies and improved molecular profiling, is strengthening drug development and expanding treatment possibilities for this aggressive cancer.
The Thymic Carcinoma Pipeline Analysis Report by Expert Market Research gives comprehensive insights into thymic carcinoma therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for thymic carcinoma. The thymic carcinoma report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The thymic carcinoma pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with thymic carcinoma treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to thymic carcinoma.
Read more about this report - Request a Free Sample
Thymic carcinoma is a rare and aggressive malignancy that develops from the thymic epithelium, a vital component for T-lymphocyte maturation. The disease arises from cellular atypia and uncontrolled proliferation within the thymus, often detected at advanced stages. Patients with unresectable tumors generally face poor prognosis due to limited treatment options. This carcinoma is exceptionally uncommon, with an estimated annual incidence of 0.29 per 100,000 individuals in Japan, highlighting the urgent need for effective therapeutic strategies and novel interventions in managing this life-threatening condition.
Thymic carcinoma treatments encompass surgery, chemotherapy, radiation therapy, and targeted approaches. Recently, immunotherapy has emerged as a promising option for unresectable or advanced cases. In December 2025, Chugai Pharmaceutical received regulatory approval in Japan for Tecentriq (atezolizumab) combined with carboplatin and paclitaxel as first-line therapy for unresectable thymic carcinoma. This approval, supported by the Phase II MARBLE study, represents the first immune-checkpoint inhibitor indicated for this rare disease, offering a significant advancement in the thymic carcinoma drug pipeline and addressing a critical unmet medical need.
The pipeline is expected to evolve with increasing research and targeted therapies. According to Zishan Chen et al., 2025, thymic epithelial tumors (TETs) showed an incidence of 2.769 and mortality of 1.203 per million person-years from 2000 to 2020. Tiemo Sven Gerber et al., 2024, reported thymic carcinoma incidence of 0.48 and 0.42 per million inhabitants. Radjab Fatmasari et al., 2025, noted thymoma incidence of 2.2 per million in the United States and 2.64 per million in Germany. In Japan, So Takata, 2024, estimated 0.15-0.29 per 100,000 person-years. These epidemiological insights support focused pipeline development.
This section of the report covers the analysis of thymic carcinoma drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
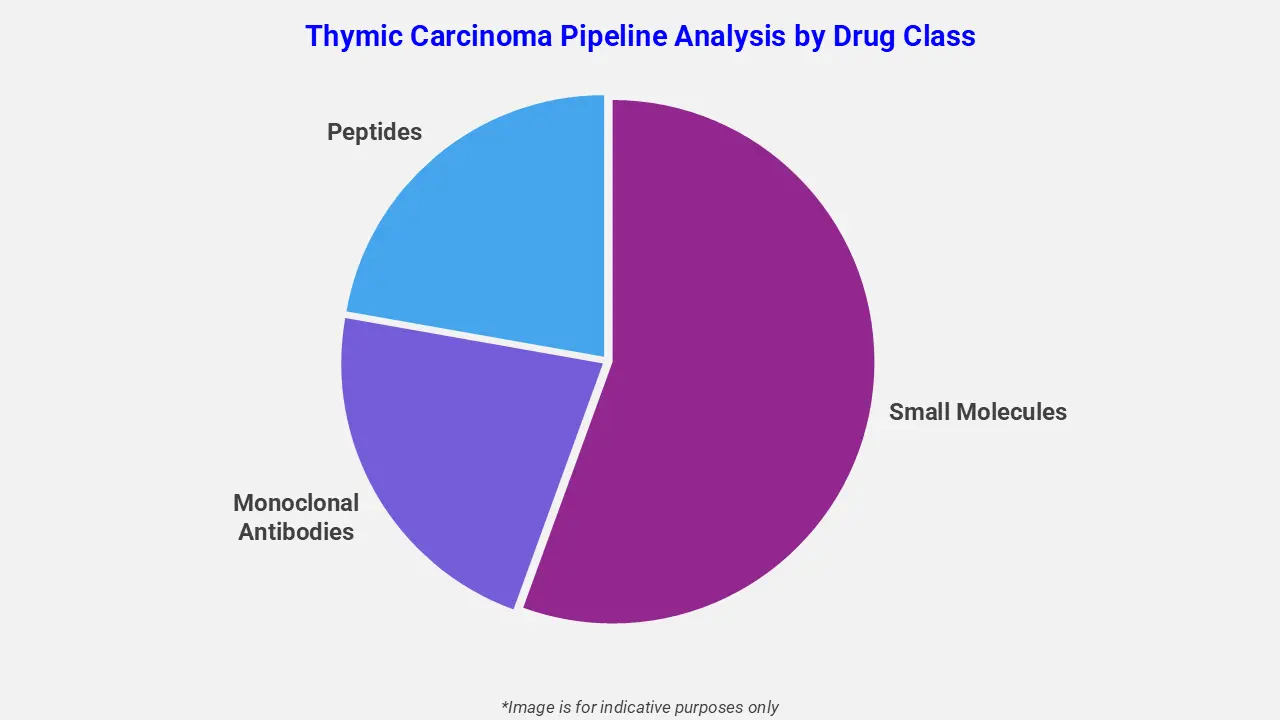
By Drug Class
The thymic carcinoma pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with a 70% share, covers a major share of the total thymic carcinoma clinical trials, reflecting high clinical advancement and strong mid‑stage development momentum. Phase I holds 20%, indicating early innovation and promising preclinical foundations. Phase III accounts for 10%, representing near‑term regulatory potential and emerging market readiness.
The drug molecule categories covered under the thymic carcinoma pipeline analysis include small molecules, monoclonal antibodies, and peptides. The thymic carcinoma report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for thymic carcinoma. Antibody-drug conjugates are gaining traction in the thymic carcinoma drug pipeline to address high unmet clinical needs. For example, HLX43, a programmed death-ligand 1 targeting ADC, is under investigation for advanced thymic epithelial tumors. It combines immune checkpoint blockade with cytotoxic payload delivery, demonstrating promising efficacy and manageable safety in later-line Thymic carcinoma patients, potentially expanding treatment options.
The EMR report for the thymic carcinoma pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed thymic carcinoma therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in thymic carcinoma clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for thymic carcinoma. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of thymic carcinoma drug candidates.
PT-112 is a first-in-class metallo-pyrophosphate conjugate being evaluated in a Phase II clinical trial sponsored by the National Cancer Institute. The study examines its efficacy and safety in patients with recurrent or refractory thymoma and thymic carcinoma, aiming to determine tumor response and shrinkage. PT-112 is a novel immunogenic cell death (ICD)-inducing small molecule that selectively inhibits cancer cell growth while sparing non-tumor cells. It activates caspase-3, triggers mitochondrial stress including ROS generation, compromises membrane integrity, alters respiration, induces autophagy, and releases damage-associated molecular patterns (DAMPs), enhancing anti-tumor immunity. The drug is administered intravenously in 28-day cycles, and the study is expected to reach primary completion in December 2026, with overall completion by June 2027.
Bintrafusp Alfa (M7824) is a first-in-class bifunctional fusion protein combining a human IgG1 anti-PD-L1 antibody with two TGF-β receptor II molecules, acting as a TGF-β trap to inhibit tumor-induced immunosuppression. Administered intravenously every two weeks, it is designed to enhance immune responses against relapsed or refractory thymoma and thymic carcinoma. Sponsored by the National Cancer Institute (NCI), this Phase II study is examining the drug’s objective response rate, safety, and immune-related effects, including tumor and blood biomarker changes. The trial is active but not recruiting, with completion expected by December 2026.
CD70-Targeted CAR-T Therapy is a novel chimeric antigen receptor T-cell (CAR-T) immunotherapy designed to target CD70-positive advanced solid tumors, including thymic carcinoma. Sponsored by Chongqing Precision Biotech Co., Ltd, this Phase 1 study is examining the safety, efficacy, and pharmacokinetics of the drug while determining the optimal dose and infusion schedule. The therapy involves engineering patient T cells with CD70-specific receptors, enhancing tumor recognition, in vivo expansion, and antitumor immune responses. Patients receive therapy via intravenous, intrapleural, or intraperitoneal infusion. The study started in September 2025 and is expected to be completed by May 2028, enrolling approximately 90 participants.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Thymic Carcinoma Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for thymic carcinoma. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into thymic carcinoma collaborations, regulatory environments, and potential growth opportunities.
Neuroendocrine Carcinoma Market




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share