
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Ventricular hypertrophy represents a significant cardiovascular condition characterized by thickening of the heart’s ventricular walls, contributing to increased morbidity and mortality worldwide. Globally, left ventricular hypertrophy affects 15-20% of adults. Growing understanding of underlying genetic and pathological mechanisms has fueled innovative drug development. According to ventricular hypertrophy pipeline analysis by Expert Market Research, ongoing clinical programs focus on cardiac myosin inhibitors, gene therapies, and RNA-based approaches to reduce ventricular remodeling and improve cardiac function.
Major companies involved in the ventricular hypertrophy pipeline analysis include Rocket Pharmaceuticals Inc., Novartis and others.
Leading drugs currently in the pipeline include Venglustat (GZ402671) and others.
Growing understanding of genetic cardiomyopathies and sarcomere dysfunction is accelerating targeted therapy development for ventricular hypertrophy. The emergence of cardiac myosin inhibitors and gene-based therapies is driving pipeline expansion, particularly for patients with hypertrophic cardiomyopathy, while advances in precision diagnostics are enabling earlier identification of disease-causing mutations and personalized treatment strategies.
The Ventricular Hypertrophy Pipeline Analysis Report by Expert Market Research gives comprehensive insights into ventricular hypertrophy therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for ventricular hypertrophy. The ventricular hypertrophy report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The ventricular hypertrophy pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with ventricular hypertrophy treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to ventricular hypertrophy.
Read more about this report - Request a Free Sample
The ventricular hypertrophy pipeline outlook is evolving as research shifts toward therapies that target the underlying mechanisms of myocardial thickening rather than only managing symptoms. Conventional management typically includes beta-blockers, calcium channel blockers, and surgical interventions such as septal reduction therapy to relieve obstruction and improve cardiac function. However, innovative pharmacological approaches are emerging within the clinical pipeline. For instance, in December 2025, the U.S. FDA approved Myqorzo (aficamten), a cardiac myosin inhibitor developed by Cytokinetics, for adults with symptomatic obstructive hypertrophic cardiomyopathy. The therapy works by reducing excessive cardiac contractility, thereby improving functional capacity and alleviating symptoms associated with ventricular wall thickening, marking a significant advancement in targeted treatment strategies.
Ventricular hypertrophy encompasses both left and right ventricular thickening, with distinct epidemiological patterns. Left ventricular hypertrophy (LVH) affects approximately 10-20% of the general adult population, rising to 30-40% among hypertensive individuals. While global prevalence of combined ventricular hypertrophy is not widely reported, the condition contributes substantially to cardiovascular morbidity and mortality. Early detection through imaging and management of underlying risk factors remains critical to reducing long‑term complications.
This section of the report covers the analysis of ventricular hypertrophy drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
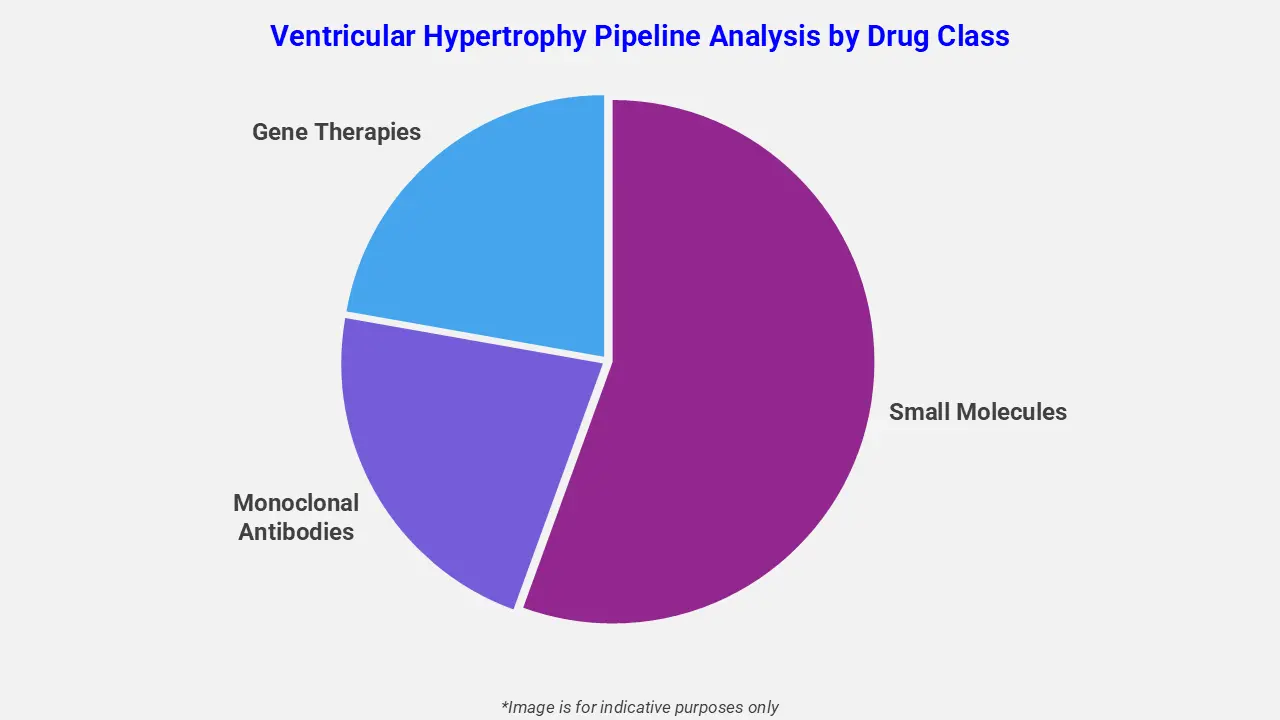
By Drug Class
The ventricular hypertrophy pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total ventricular hypertrophy clinical trials. Phase II accounts for 60%, followed by phase IV (20%), early phase I (10%), and phase III (10%), highlighting Phase II as the dominant stage.
The drug molecule categories covered under the ventricular hypertrophy pipeline analysis include small molecules, monoclonal antibodies, and gene therapies. The ventricular hypertrophy report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for ventricular hypertrophy. For instance, in June 2023, the FDA approved an update to the label of Camzyos (mavacamten), incorporating additional clinical data from the Phase III VALOR-HCM study. The drug, developed by Bristol Myers Squibb, is a first-in-class cardiac myosin inhibitor that targets the underlying sarcomere dysfunction responsible for ventricular hypertrophy in obstructive hypertrophic cardiomyopathy. The updated label further strengthened its clinical evidence for improving symptoms and functional capacity in affected patients.
The EMR report for the ventricular hypertrophy pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed ventricular hypertrophy therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in ventricular hypertrophy clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for ventricular hypertrophy. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of ventricular hypertrophy drug candidates.
Venglustat (GZ402671) is an investigational small-molecule glucosylceramide synthase inhibitor being developed by Sanofi. It belongs to the substrate reduction therapy class and is being studied in patients with Fabry disease–associated ventricular hypertrophy. The drug works by inhibiting glucosylceramide synthase, thereby reducing the production of glycosphingolipids that accumulate in cardiac tissue and contribute to pathological cardiac remodeling and ventricular wall thickening. A Phase III clinical study is evaluating its ability to improve left ventricular mass index, a key indicator of hypertrophic cardiac involvement. Sanofi is leading the global clinical development program to assess its long-term cardiovascular benefits.
CDR132L is an investigational antisense oligonucleotide RNA-based therapy developed by Cardior Pharmaceuticals for cardiovascular diseases associated with ventricular hypertrophy. The therapy targets and inhibits microRNA-132, a regulatory molecule that drives pathological cardiac hypertrophy and adverse remodeling of heart muscle cells. By blocking abnormal miR-132 signaling, CDR132L aims to restore normal cardiomyocyte function, reduce hypertrophic growth, and improve cardiac contractility. Clinical studies have demonstrated promising effects on cardiac function and biomarkers in heart failure models. Cardior Pharmaceuticals is advancing the drug through clinical trials, with further development supported by Novo Nordisk following its acquisition of the company.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Ventricular Hypertrophy Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for ventricular hypertrophy. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into ventricular hypertrophy collaborations, regulatory environments, and potential growth opportunities.
Hypertrophic Cardiomyopathy Epidemiology Forecast




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share