
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Advanced cervical cancer remains a critical public health concern, particularly in regions with limited access to screening and timely treatment. Globally, cervical cancer accounts for approximately 660,000 new cases annually, with a substantial proportion diagnosed at advanced stages due to delayed detection. Mortality remains high in low- and middle-income countries, reflecting care disparities. According to the advanced cervical cancer pipeline analysis by Expert Market Research, rising late-stage diagnoses and therapeutic innovation are shaping future clinical development and competitive positioning.
Major companies involved in the advanced cervical cancer pipeline analysis include AstraZeneca, Evopoint Biosciences Inc., and others.
Leading drugs currently in the pipeline include XNW28012, AK104, and others.
The advanced cervical cancer pipeline is driven by expanding use of antibody-drug conjugates, tumor-infiltrating lymphocyte therapies, and precision immunotherapies targeting HPV-related oncogenic pathways. Increasing biomarker stratification, rising incidence in younger women, and unmet needs after platinum failure are accelerating innovation, strategic licensing deals, and regulatory fast-track designations globally.
The Advanced Cervical Cancer Pipeline Analysis Report by Expert Market Research gives comprehensive insights into advanced cervical cancer therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for advanced cervical cancer. The advanced cervical cancer report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The advanced cervical cancer pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with advanced cervical cancer treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to advanced cervical cancer.
Read more about this report - Request a Free Sample
The advanced cervical cancer pipeline outlook reflects growing innovation aimed at improving outcomes beyond standard platinum-based chemotherapy, radiation therapy, and immune checkpoint inhibitors used in recurrent or metastatic settings.
Advanced cervical cancer treatment continues to evolve as resistance and relapse after standard regimens highlight significant unmet clinical need. While platinum-based chemotherapy, radiation, and immunotherapy remain foundational, durable responses are still limited in advanced stages. For instance, in January 2024, the FDA approved pembrolizumab in combination with chemoradiotherapy for FIGO Stage III-IVA cervical cancer, marking a major advancement and expanding treatment options for patients with locally advanced disease who historically had limited effective therapies.
Globally, cervical cancer is the fourth most common cancer among women, with an estimated 660,000 new cases and approximately 350,000 deaths in 2022, disproportionately affecting low- and middle-income countries where access to prevention and treatment is limited. Persistent infection with high-risk human papillomavirus (HPV) is the primary cause, and women living with HIV are about six times more likely to develop the disease. Regional disparities in incidence and mortality reflect inequities in vaccination, screening, and treatment services, underscoring the need for strengthened global health efforts.
This section of the report covers the analysis of advanced cervical cancer drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
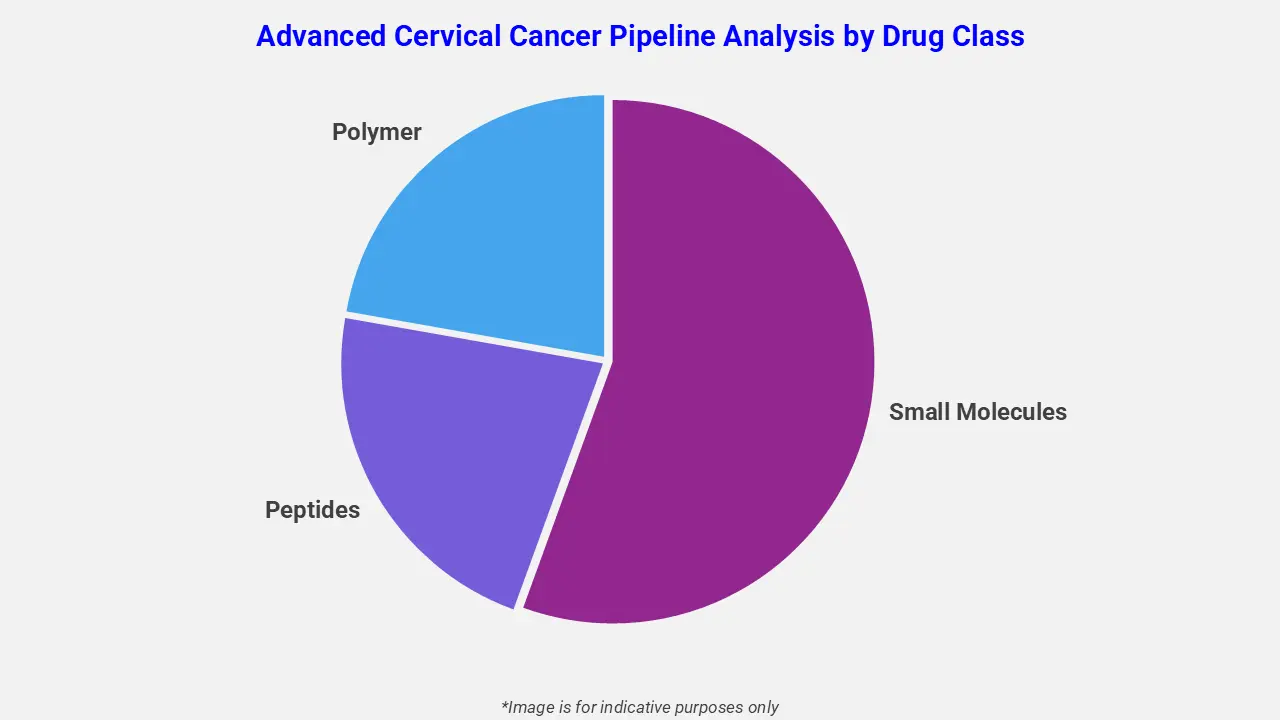
By Drug Class
The advanced cervical cancer pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total advanced cervical cancer clinical trials. The report covers phase I (35%), phase II (52%), and phase III (12%) drugs in global advanced cervical cancer clinical trials.
The drug molecule categories covered under the advanced cervical cancer pipeline analysis include small molecules, peptides, and polymers. The advanced cervical cancer report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for advanced cervical cancer. For instance, in April 2024, the FDA granted full approval to tisotumab vedotin-tftv (Tivdak), an antibody-drug conjugate, for recurrent or metastatic cervical cancer after prior chemotherapy, reinforcing the growing emphasis on precision-driven therapeutic strategies.
The EMR report for the advanced cervical cancer pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed advanced cervical cancer therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in advanced cervical cancer clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for advanced cervical cancer. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of advanced cervical cancer drug candidates.
PVX7 is a therapeutic prime/boost HPV vaccine developed by PapiVax Biotech, Inc. designed to treat HPV-associated cancers, including advanced cervical cancer, by stimulating the immune system to target viral oncoproteins E6 and E7. It combines a DNA vaccine (pBI-11) with a recombinant vaccinia vector (TA-HPV) to elicit cytotoxic T-cell responses against tumor cells expressing HPV16/18 antigens. PVX7 is being evaluated in clinical studies at leading centers such as Johns Hopkins and the Sidney Kimmel Comprehensive Cancer Center, with support from NIH collaborators, reflecting ongoing academic-industry efforts in immunotherapy.
XNW28012 is an antibody-drug conjugate (ADC) co-developed by Shanghai Sinovent Biopharmaceutical and advanced clinically by Evopoint Biosciences Inc. that targets specific tumor antigens to deliver cytotoxic agents directly into cancer cells. ADCs leverage a monoclonal antibody linked to a potent drug, aiming to inhibit tumor growth effectively while limiting systemic toxicity. In early-phase clinical trials, XNW28012 is being tested in patients with advanced solid tumors, including cervical cancer, who have exhausted standard therapies, demonstrating the developer’s commitment to expanding options for difficult-to-treat malignancies.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Advanced Cervical Cancer Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for advanced cervical cancer. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into advanced cervical cancer collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share