
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Brain hemorrhage, or intracerebral hemorrhage (ICH), is a severe condition caused by bleeding within the brain tissue, often due to trauma, hypertension, or aneurysm rupture. It leads to increased intracranial pressure, brain edema, and disrupted neural function. Symptoms include sudden headache, weakness, and altered consciousness. Treatment focuses on stabilizing the patient, controlling bleeding, and reducing pressure through medications or surgery. Prompt intervention is critical to minimize neurological damage and improve outcomes. The brain hemorrhage pipeline analysis by Expert Market Research focuses on various treatment options for this disease.
Overview: For companies and buyers tracking the Brain Hemorrhage Pipeline Insight sector, Q1 2026 was a quarter unlike any recent precedent. Environment for Brain Hemorrhage drug pipeline development, with defense medical investment elevating certain therapeutic priorities, supply chain disruption affecting clinical trial materials and API logistics, and geopolitical risk adding new dimensions to clinical development strategy. The conflict's medical countermeasure requirements, particularly for trauma care, CBRN therapeutics, and infectious disease, generated accelerated development pathways that intersect with commercial Brain Hemorrhage pipeline programs. BARDA and DoD emergency use authorization precedents from Q1 2026 are informing regulatory strategy for expedited pipeline programs.
United States: US Brain Hemorrhage pipeline activity during the first quarter of 2026 was sustained by FDA's operational continuity and the pharmaceutical industry's established contingency protocols for clinical trial management during the geopolitical turmoil. US defense medical investment, with BARDA funding elevated by Q1 2026 threat environment, created additional development program funding for Brain Hemorrhage therapeutic areas with dual defense-civilian application. US pharmaceutical companies advancing Brain Hemorrhage pipeline programs navigated API and clinical supply logistics disruption from Hormuz closure, with some India-manufactured API supply chains affected by elevated shipping costs and lead time extension from Q1 2026 logistics disruption. Major US pharma investors including NIH, NCI, and BARDA maintained funding commitments for Brain Hemorrhage pipeline programs through Q1 2026.
Iran & Israel: Israeli pharmaceutical and biotech companies advancing Brain Hemorrhage pipeline programs, including Teva Pharmaceutical, Kamada, and numerous early-stage biotech companies in Israel's deep life science environment, maintained research and development operations throughout Q1 2026 conflict conditions. Israel's medtech and pharma sector demonstrated wartime resilience, with R&D programs, clinical trial management, and regulatory submissions continuing without material disruption in most cases. Gulf state pharmaceutical investment, supported by elevated oil revenues from Brent crude at USD 126/barrel (Wikipedia) , maintained Vision 2030 healthcare localization and pharmaceutical manufacturing investment programs including Brain Hemorrhage-relevant therapeutic development partnerships. Iranian pharmaceutical pipeline programs, already severely constrained by sanctions limiting access to clinical trial materials, international collaboration, and regulatory pathway engagement, faced additional disruption from Q1 2026 conflict impacts on research infrastructure.
Government & Policy Watch
Market & Industry Impact
Procurement & Supply Chain Alert
Major companies involved in the brain hemorrhage pipeline analysis include Bioxodes S.A., Grace Therapeutics Inc., and Penumbra Inc., among others.
Leading drugs currently in the pipeline include Ir-CPI, GTX-104, and others.
Increased investment in research and development, along with regulatory support, is accelerating clinical trials and new treatment approvals.
The Brain Hemorrhage Pipeline Analysis Report by Expert Market Research gives comprehensive insights into brain hemorrhage therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for Brain Hemorrhage therapeutics. The brain hemorrhage report assessment includes the analysis of over 15 pipeline drugs and 10+ companies. The brain hemorrhage pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with brain hemorrhage treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to brain hemorrhage.
Read more about this report - REQUEST FREE SAMPLE COPY IN PDF
Brain hemorrhage is caused by a combination of primary and secondary damage processes. Primary injury results from blood vessel rupture, which raises intracranial pressure, reduces oxygen delivery, and forms hematomas. Secondary damage is caused by iron toxicity from blood breakdown, oxidative stress, and inflammation. It occurs over the course of days to weeks. Brain injury and neurological abnormalities are exacerbated by these processes, which also produce edema, rupture the blood-brain barrier, and cause neuronal death.
Treatment of brain hemorrhage focuses on stopping bleeding, reducing intracranial pressure, and preventing complications. Emergency measures include medications to control blood pressure, reverse anticoagulants, and reduce brain swelling. Surgical interventions like craniotomy or minimally invasive procedures are used to remove hematomas, repair ruptured vessels, or relieve pressure. Long-term rehabilitation includes physical, speech, and occupational therapy to restore function. Timely treatment is critical for improving survival and minimizing neurological damage
Brain hemorrhages make up 10–20% of all strokes worldwide, and they are more common in low- and middle-income nations, where incidence rates are 22 per 100,000 person-years, as opposed to 10 in high-income areas. The burden is greater in men, older persons, and those with vascular abnormalities or hypertension. East Asia and Sub-Saharan Africa have a disproportionately high rate of intracerebral hemorrhages, which is indicative of differences in risk factor management and healthcare availability.
This section of the report covers the analysis of brain hemorrhage drug candidates based on several segmentations, including:
By Phase
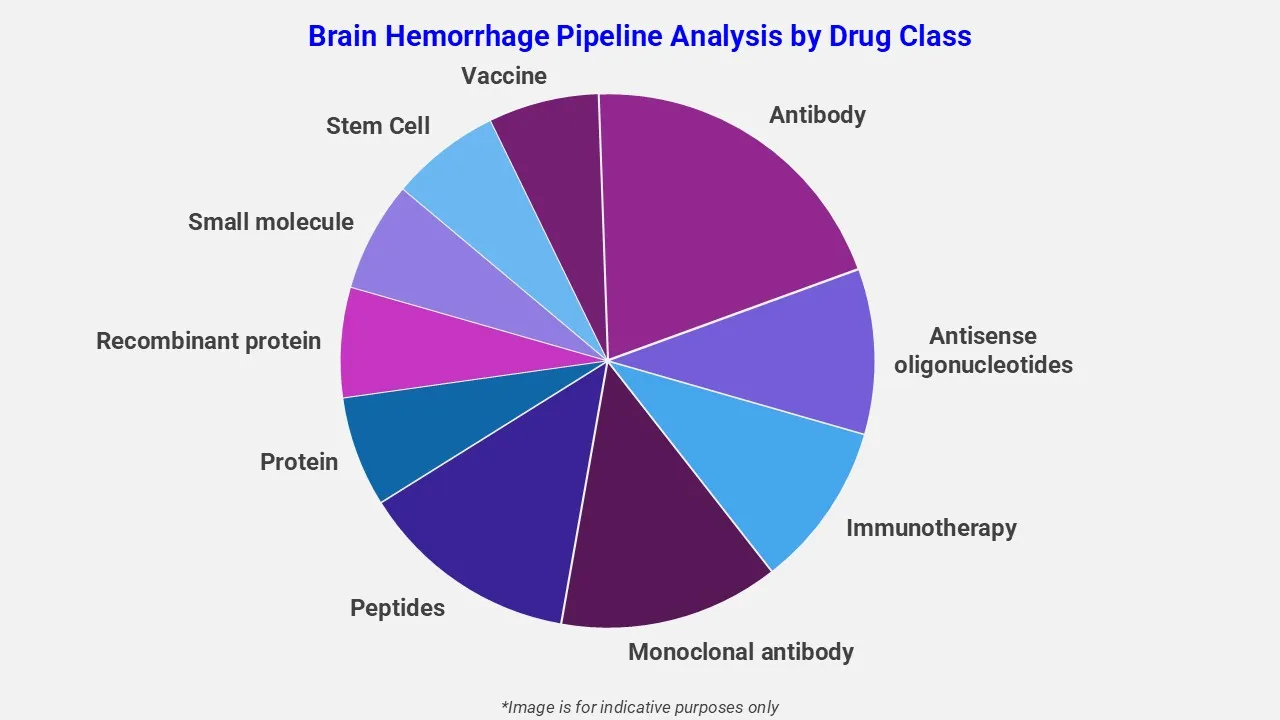
By Drug Class
By Route of Administration
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total brain hemorrhage clinical trials.
In the brain hemorrhage pipeline, most of the candidates are concentrated in Phase II and Phase III with 50% of the proposed drugs in each. Thus, demonstrating a broad spectrum of development stages and diverse progress toward potential treatments.
The drug molecule categories covered under the brain hemorrhage pipeline analysis include monoclonal antibodies, peptides, small molecules and gene therapy. The brain hemorrhage report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for brain hemorrhage.
The EMR report for the brain hemorrhage pipeline analysis covers the profile of key companies involved in clinical trials and their drugs under development. Below is the list of a few players involved in brain hemorrhage clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for brain hemorrhage. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of brain hemorrhage drug candidates.
Ir-CPI (BIOX-101), developed by Bioxodes S.A., is a novel biologic derived from the salivary protein of the Ixodes ricinus tick. It inhibits coagulation factors XIa and XIIa to prevent thrombosis without increasing bleeding risk. Ir-CPI also reduces neuroinflammation by limiting neutrophil activation and NET release. It is currently undergoing Phase II clinical trials to assess its safety and efficacy in patients with intracerebral hemorrhage (ICH).
GTX-104, developed by Grace Therapeutics Inc., is an innovative intravenous formulation of nimodipine designed to treat aneurysmal subarachnoid hemorrhage (aSAH). This novel formulation aims to overcome limitations associated with oral nimodipine, such as poor bioavailability and dosing challenges in unconscious patients. The pivotal Phase 3 STRIVE-ON trial demonstrated that GTX-104 met its primary endpoint, showing a 19% reduction in clinically significant hypotension episodes compared to oral nimodipine. These results support its potential as a safer and more effective treatment for aSAH.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
Brain Hemorrhage Epidemiology Forecast




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.