
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Fallopian tube cancer is a rare gynecologic malignancy, with only 1,500 to 2,000 cases reported worldwide and an estimated 300 to 400 new diagnoses annually in the United States. The disease often develops quietly and may spread to nearby pelvic organs or distant sites before detection. Treatment approaches for fallopian tube cancer typically include surgery, chemotherapy, targeted therapy, and, increasingly, immunotherapy as precision oncology advances. The growing focus on novel therapeutic agents and molecularly driven strategies is transforming the management landscape. According to the fallopian tube cancer pipeline analysis by Expert Market Research, ongoing research and emerging candidates indicate a steadily expanding pipeline and notable opportunities for future clinical development.
Major companies involved in the fallopian tube cancer pipeline include AstraZeneca, AbbVie, and several other leading biopharmaceutical innovators that are actively advancing targeted therapies, antibody-drug conjugates, and immuno-oncology approaches to address this rare but clinically challenging malignancy.
Leading drugs currently in the pipeline include Mirvetuximab soravtansine plus Bevacizumab, LY4170156, and other emerging candidates designed to improve treatment precision, enhance response rates, and offer new therapeutic options beyond conventional chemotherapy.
The pipeline is further driven by rising diagnostic awareness, advancements in molecular profiling, and the growing emphasis on developing innovative treatments for rare gynecologic cancers, which collectively support strong research momentum and future market growth in fallopian tube cancer.
The Fallopian Tube Cancer Pipeline Analysis Report by Expert Market Research gives comprehensive insights into fallopian tube cancer therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for fallopian tube cancer. The fallopian tube cancer report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The fallopian tube cancer pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with fallopian tube cancer treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to fallopian tube cancer.
Read more about this report - Request a Free Sample
Fallopian tube cancer treatment typically involves surgery, chemotherapy, targeted therapies, and, in select cases, immunotherapy to control tumor growth, reduce recurrence risk, and improve survival outcomes in both early and advanced disease. Platinum-based regimens remain the cornerstone of therapy, often combined with agents such as bevacizumab to enhance clinical response. Recent advancements in antibody-drug conjugates and PARP inhibitors are expanding treatment options, particularly for patients with BRCA mutations or homologous recombination deficiency. Ongoing clinical trials evaluating combinations like mirvetuximab soravtansine with bevacizumab highlight emerging strategies that aim to establish more effective standards of care in fallopian tube cancer.
The global burden of fallopian tube cancer remains clinically significant, with only 1,500 to 2,000 cases reported worldwide, underscoring its rarity among gynecologic malignancies. In the United States, an estimated 300 to 400 new diagnoses occur annually, according to UCSF Health, reflecting its limited incidence but challenging diagnostic profile. Although comprehensive global datasets are fewer due to its rarity, emerging clinical reports suggest increased recognition of the disease as diagnostic methods improve, and awareness grows among clinicians. These epidemiological insights highlight the need for continued research and development of targeted therapies to improve outcomes in fallopian tube cancer.
This section of the report covers the analysis of fallopian tube cancer drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
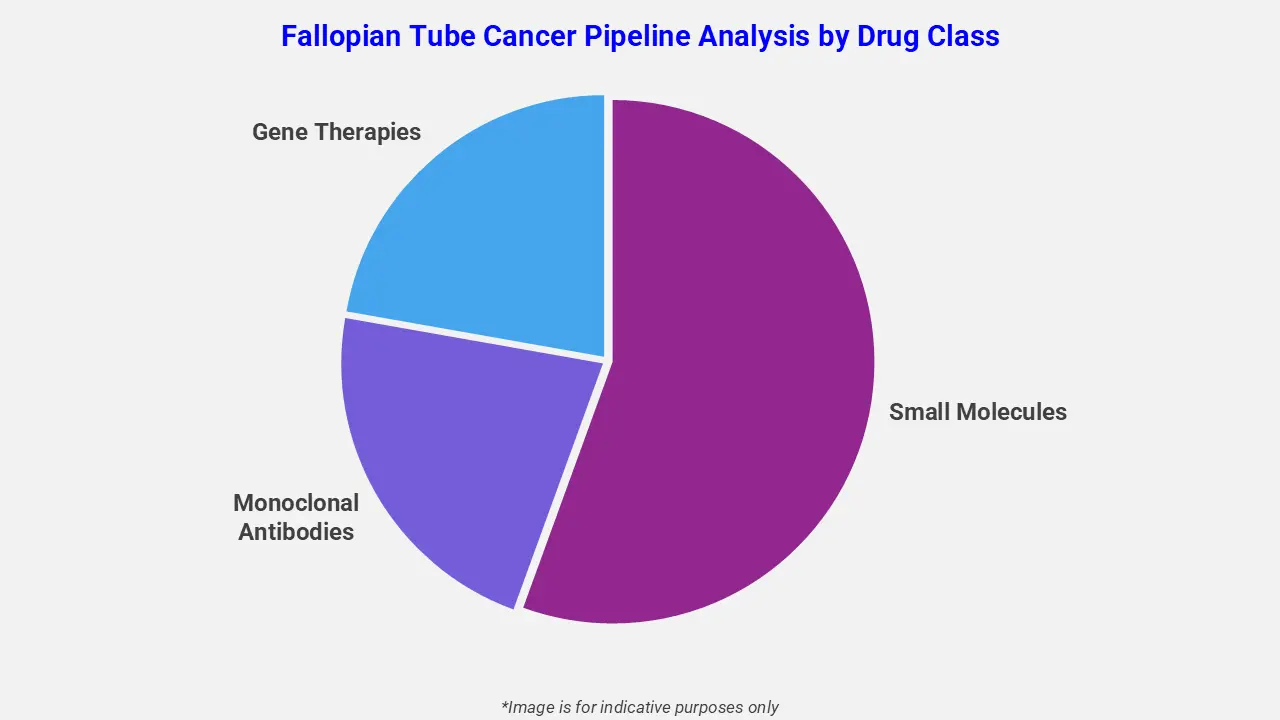
By Drug Class
The fallopian tube cancer pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with 58.5%, covers a major share of the total fallopian tube cancer clinical trials. It is followed by phase I at 34%. The strong representation in Phase I and II highlights robust clinical development, signaling potential approvals, increased treatment options, and growth opportunities in the market.
The drug molecule categories covered under the fallopian tube cancer pipeline analysis include small molecules, monoclonal antibodies, and gene therapies. The fallopian tube cancer report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for fallopian tube cancer. Recent developments highlight growing interest in advanced biologics, particularly antibody-drug conjugates targeting folate receptor-α, which have shown promising activity in fallopian tube cancer. Ongoing trials evaluating combinations such as mirvetuximab soravtansine with bevacizumab reflect the shift toward more precise, mechanism-driven therapies. These innovations underscore the expanding therapeutic landscape and the increasing focus on improving outcomes in this rare gynecologic malignancy.
The EMR report for the fallopian tube cancer pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed fallopian tube cancer therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in fallopian tube cancer clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for fallopian tube cancer. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of fallopian tube cancer drug candidates.
Mirvetuximab soravtansine combined with Bevacizumab is being evaluated in a Phase III study for maintenance therapy in platinum-sensitive epithelial ovarian, fallopian tube, or peritoneal cancer. Sponsored by AbbVie in collaboration with the GOG Foundation, the trial aims to determine whether the combination improves progression-free outcomes compared with Bevacizumab alone in patients responding to platinum-based chemotherapy. Mirvetuximab soravtansine, an antibody-drug conjugate targeting folate receptor-α, delivers a cytotoxic payload directly to cancer cells, while Bevacizumab inhibits angiogenesis. The study is recruiting, with an expected start in March 2027 and completion in April 2029, assessing efficacy, safety, and durability of response.
Bevacizumab is an anti-angiogenic monoclonal antibody being evaluated in a Phase II study for ovarian, fallopian tube, primary peritoneal cancer, and other malignant neoplasms of the female genital tract in patients undergoing second-look surgery. Sponsored by M.D. Anderson Cancer Center in collaboration with the National Cancer Institute (NCI), the study examines whether Bevacizumab can reduce residual disease and delay recurrence by inhibiting vascular endothelial growth factor (VEGF), thereby suppressing tumor angiogenesis. Although active but not recruiting, the trial focuses on assessing safety, therapeutic activity, and surgical outcomes, with both study start and completion set for February 1, 2026.
Cirtuvivint combined with Olaparib is being investigated in a Phase I study for platinum-resistant ovarian cancer with BRCA mutations or homologous recombination deficiency, including endometrioid ovarian, primary peritoneal, and fallopian tube cancers. Sponsored by the University of Colorado, Denver, in collaboration with the NIH and National Cancer Institute (NCI), the trial aims to determine whether dual inhibition of Wnt/β-catenin signaling (via Cirtuvivint) and DNA repair mechanisms (via Olaparib) enhances antitumor activity in heavily pretreated patients. Currently recruiting, the study will assess safety, dose optimization, pharmacokinetics, and preliminary efficacy, with an expected start in July 2027 and completion in July 2029.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Fallopian Tube Cancer Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for Fallopian Tube Cancer. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into fallopian tube cancer collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share