
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Lung adenocarcinoma, a prominent form of non-small cell lung cancer (NSCLC), originates in glandular epithelial cells located in the outer regions of the lungs. According to the World Health Organization data, there were over 2.4 million new lung cancer cases, with lung cancer in non-smokers and women increasingly contributing to the rising share of adenocarcinoma cases. Current pipeline innovations are largely centered on precision oncology in lung cancer, including targeted cancer therapies and lung cancer immunotherapy, aiming to surpass the limitations of conventional chemotherapy and surgical approaches. According to the lung adenocarcinoma pipeline analysis by Expert Market Research, various pipeline candidates are under development, highlighting rapid advancements in lung cancer drug development. The growing disease burden, coupled with continuous innovation in biomarker-driven therapies and increased R&D investments, is expected to significantly transform the lung adenocarcinoma treatment landscape over the forecast period.
Major companies involved in the lung adenocarcinoma pipeline analysis include Sichuan Baili Pharmaceutical Co., Ltd., Lantern Pharma Inc., and others.
Leading drugs currently in the pipeline include SI-B001 + Docetaxel, LP-300 + Pemetrexed + Carboplatin, PF-07220060, and others.
The pipeline is witnessing expansion through EGFR inhibitors, KRAS mutation therapies, and advancements in immune checkpoint inhibitors, with a growing emphasis on combination cancer therapies and personalized treatment strategies.
The Lung Adenocarcinoma Pipeline Analysis Report by Expert Market Research gives comprehensive insights into lung adenocarcinoma therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for lung adenocarcinoma. The lung adenocarcinoma report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The lung adenocarcinoma pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with lung adenocarcinoma treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to lung adenocarcinoma.
Read more about this report - Request a Free Sample
Lung adenocarcinoma, a major subtype of NSCLC, develops due to genetic alterations, environmental exposures such as smoking and air pollution, and hereditary risk factors. These triggers contribute to abnormal cellular proliferation, tumorigenesis, and metastasis, underscoring the importance of early diagnosis and molecular testing in lung cancer.
Lung adenocarcinoma treatments include surgical intervention, chemotherapy, targeted therapy for lung cancer, immuno-oncology treatments, and radiation therapy, all tailored according to disease stage and genetic profile. In December 2024, zenocutuzumab-zbco (Bizengri, Merus N.V.) was approved by the FDA for NRG1 fusion-positive advanced or metastatic non-small cell lung cancer, demonstrating a 33% overall response rate and providing a novel systemic option for patients with disease progression after prior therapies.
Lung adenocarcinoma, a subtype of non-small cell lung cancer, remains a leading cause of cancer-related mortality. According to David J. Myers et al., 2023, it accounts for approximately 40% of all lung cancers in the United States and is strongly associated with prior smoking. As per the World Health Organization and IARC’s Global Cancer Observatory, lung cancer was the most common cancer globally in 2022, with 2.5 million new cases representing 12.4% of all cancers. The American Cancer Society projects 234,580 new lung cancer cases and 125,070 deaths in the United States in 2024, reinforcing the urgency for advancements in lung cancer early detection and treatment innovation.
This section of the report covers the analysis of lung adenocarcinoma drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
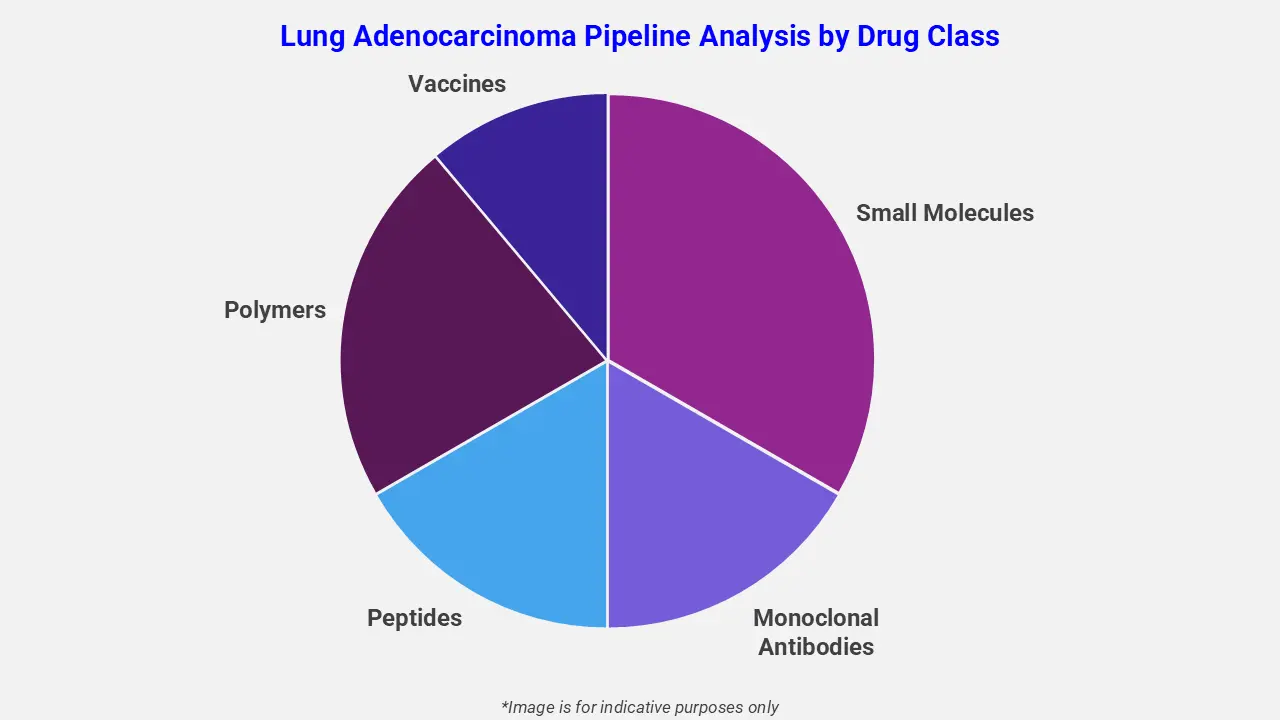
By Drug Class
The lung adenocarcinoma pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase I, with 51%, covers a major share of the total lung adenocarcinoma clinical trials, driven by high initiation of novel compounds and early safety profiling. Phase II, with 43%, reflects strong efficacy testing and patient recruitment. Phase III holds nearly 5% benefits from late-stage validation and regulatory readiness. Overall, this distribution strengthens pipeline robustness and accelerates market growth.
The drug molecule categories covered under the lung adenocarcinoma pipeline analysis include small molecules, monoclonal antibodies, peptides, polymers, and vaccines. The lung adenocarcinoma report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for lung adenocarcinoma. Targeted therapies are gaining momentum in the pipeline, particularly for EGFR-mutated non-small cell lung cancer. For example, Rybrevant® (amivantamab) in combination with platinum-based chemotherapy is under investigation to improve progression-free survival in patients resistant to osimertinib. This bispecific antibody selectively targets EGFR and MET mutations, inhibiting tumor growth, and enhancing anti-tumor activity.
The EMR report for the lung adenocarcinoma pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed lung adenocarcinoma therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in lung adenocarcinoma clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for Lung Adenocarcinoma. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of lung adenocarcinoma drug candidates.
SI-B001, developed by Sichuan Baili Pharmaceutical Co., Ltd., is a bispecific antibody designed to simultaneously target the EGFR and HER3 signaling pathways, inhibiting tumor growth and proliferation in non-small cell lung adenocarcinoma. Administered intravenously in combination with docetaxel, this Phase III study examines its effect on overall survival (OS) and progression-free survival (PFS) compared to docetaxel alone. The trial is also assessing objective response rate (ORR), disease control rate (DCR), duration of response (DOR), pharmacokinetics, immunogenicity, adverse events, and patient quality of life, providing comprehensive data on safety and efficacy in second-line therapy.
LP-300 is an investigational disulfide small molecule sponsored by Lantern Pharma Inc., being evaluated in a Phase II trial in combination with carboplatin and pemetrexed for never-smoker patients with advanced lung adenocarcinoma. This study is examining progression-free survival (PFS) and overall survival (OS) to determine potential clinical advantages over standard chemotherapy alone. LP-300 works by modulating cellular redox pathways and directly engaging tyrosine kinase inhibitor (TKI) receptors through cysteine modification, targeting key signaling in non-small-cell lung cancer. Administered intravenously on day 1 of a 21-day cycle, it is being tested for safety and efficacy across 4-6 cycles, followed by possible pemetrexed maintenance.
PF-07220060, also known as Atirmociclib, is a next-generation, selective CDK4 inhibitor being developed by Pfizer. This Phase 1/2A study is actively evaluating its safety, tolerability, pharmacokinetics, and pharmacodynamics in participants with advanced solid tumors, including lung adenocarcinoma. Administered orally as a single agent and in combination with endocrine therapies (letrozole, fulvestrant) or enzalutamide, Atirmociclib works by selectively inhibiting CDK4, reducing tumor cell proliferation while minimizing effects on CDK6-related neutrophil reduction. The study examines dose escalation, maximum tolerated dose, combination therapy effects, and potential food or drug interactions to optimize therapeutic efficacy and anti-tumor responses.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Lung Adenocarcinoma Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for lung adenocarcinoma. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into lung adenocarcinoma collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|---|---|
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share