
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Parkinson’s disease continues to present a significant and expanding global health burden, driven by rising prevalence and aging populations worldwide. Current estimates indicate that over 11 million individuals are affected globally, with incidence steadily increasing across major markets. The evolving treatment landscape reflects strong innovation, particularly in gene therapies and advanced delivery systems. The Parkinson’s disease pipeline analysis by Expert Market Research highlights a robust and diversified pipeline, with increasing clinical focus on disease-modifying approaches and improved patient outcomes, positioning the market for sustained long-term therapeutic advancement.
Overview: Activity in the Parkinsons Disease Drug Pipeline Analysis space shifted measurably during Q1 2026, driven by the cascading effects of the Iran-US-Israel war. The exceptional scale of Q1 2026 combat operations, with IDF striking 500 military targets across 26 of Iran's 31 provinces (ACLED, March 2026) and US striking 8,000+ targets including 130 Iranian naval vessels (CENTCOM Admiral Brad Cooper, JNS.org), generated trauma care, emergency medical, and surgical demand that proved out advanced medical technology and accelerated defense medical procurement. The conflict at the same time drove CBRN preparedness investment, elevated pharmaceutical and medical supply chain resilience focus, and created humanitarian medical needs across conflict-affected populations. Medical supply chain disruption from Hormuz logistics constraints, with vessel traffic falling from 138/day to just 2 by March 5, 2026 (Z2Data), affected pharmaceutical raw material and medical device component supply chains that depend on Gulf-route shipping.
United States: US demand through Q1 2026 was elevated by defense medical procurement, with combat casualty care requirements from Q1 2026 operations generating stepped-up procurement of trauma products, blood management systems, surgical supplies, and emergency medical equipment through defense medical logistics channels. BARDA and DoD medical countermeasures investment, heightened by Q1 2026 CBRN threat environment from conflict with Iran's known chemical and biological weapons precursor capability, stepped-up procurement of CBRN medical countermeasures and diagnostic systems. US civilian healthcare demand for parkinsons disease drug pipeline analysis held onto structural growth driven by aging demographics, chronic disease burden, and technology advancement, with Q1 2026 providing an additional defense channel demand increment. US pharmaceutical and medical device supply chains navigated Q1 2026 raw material and component logistics disruption through inventory buffering and alternative sourcing strategies.
Iran & Israel: From an Israeli and Gulf state perspective, Israeli medical technology companies, Medigus, Given Imaging (Medtronic), Mazor Robotics (Medtronic), and the broader Israeli medtech environment, kept operating through Q1 2026, with some companies experiencing stepped-up procurement inquiries from conflict-driven demand for Israeli-developed medical technology. Gulf state healthcare investment, Saudi Arabia's Vision 2030 health sector transformation and UAE's ambitious healthcare quality improvement programs, maintained parkinsons disease drug pipeline analysis procurement supported by elevated oil revenues at Brent crude at USD 126/barrel (Wikipedia), with GCC governments prioritizing healthcare system capacity as a strategic investment. Iranian healthcare, managing both civilian and conflict-related medical needs under severe sanctions constraints, faced critical parkinsons disease drug pipeline analysis supply disruption from Q1 2026, with medical equipment and pharmaceutical access further constrained beyond pre-conflict sanctions limitations.
Major companies involved in Parkinson’s disease pipeline analysis include Serina Therapeutics, Vertero Therapeutics, and others.
Leading drugs currently in the pipeline include VGN R09b, IPS101A, BHV 8000, and others.
Advancements in targeted gene therapies and continuous drug delivery platforms are accelerating pipeline diversification, while increased biomarker-driven clinical trials are improving patient stratification and enhancing late-stage success rates.
The Parkinson’s disease Pipeline Analysis Report by Expert Market Research gives comprehensive insights into Parkinson’s disease therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for Parkinson’s Disease. The Parkinson’s disease report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The Parkinson’s disease pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with Parkinson’s disease treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to Parkinson’s Disease.
Read more about this report - REQUEST FREE SAMPLE COPY IN PDF
Parkinson’s disease is a progressive neurodegenerative disorder causing motor dysfunction, tremors, rigidity, and nonmotor symptoms, primarily affecting older adults worldwide.
Parkinson’s disease treatment primarily relies on dopaminergic therapies such as levodopa, dopamine agonists, and adjunctive agents, which help manage motor symptoms but often lead to fluctuations over time. The pipeline is expanding toward continuous delivery systems, gene therapies, and neuroprotective approaches. In October 2024, the U.S. Food and Drug Administration approved Vyalev™ (foscarbidopa and foslevodopa), a 24-hour subcutaneous infusion therapy by AbbVie, improving “on” time and reducing motor fluctuations, marking a significant advancement in managing advanced Parkinson’s disease.
Parkinson’s disease epidemiology reflects a substantial and growing global burden driven by aging populations and demographic shifts. According to the Global Burden of Disease Study 2021, approximately 11.77 million people were living with Parkinson’s disease worldwide, representing a 274% increase from 1990. As per BMJ Open 2025, 1.34 million new cases were reported globally in 2021, with incidence rates rising significantly over time. The disease predominantly affects individuals aged 65 and older, particularly those above 80. Evidence from Lancet Healthy Longevity indicates slightly higher prevalence in males, while The Lancet 2024 highlights notable ethnic disparities globally.
This section of the report covers the analysis of Parkinson’s disease drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
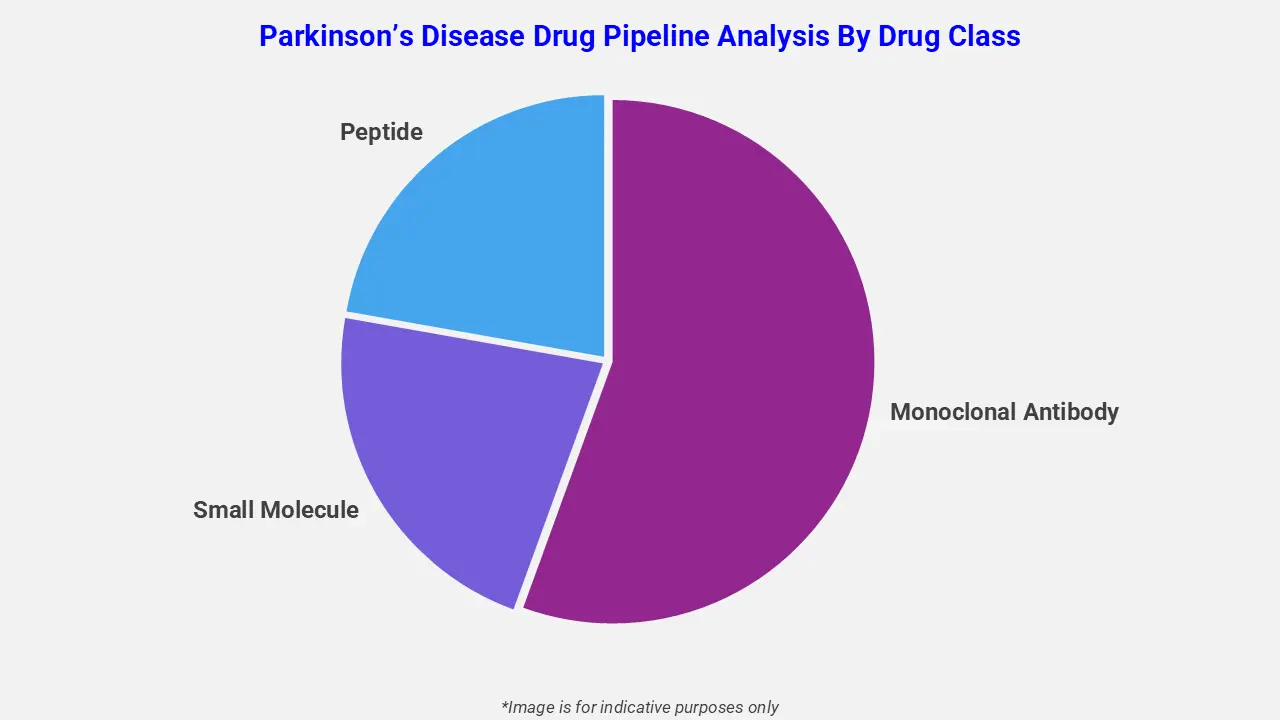
By Drug Class
The Parkinson’s disease pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total Parkinson’s disease clinical trials, commanding 42%, accelerating efficacy validation. Other phases commanded as early Phase 1 (6%), Phase 1 (30%), Phase 3 (13%), and Phase 4 (10%) drug candidates, providing an in-depth analysis of each. Overall, this phase-wise distribution reflects a well-balanced and advancing pipeline, indicating sustained innovation and a strong trajectory toward improved Parkinson’s Disease treatment outcomes.
The drug molecule categories covered under the Parkinson’s disease pipeline analysis include small molecules, oligonucleotides, and peptides. The Parkinson’s disease report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for Parkinson’s Disease. Novel drug classes for Parkinson’s Disease are advancing to address drug-resistant strains and improve treatment outcomes. For instance, in February 2025, the FDA approved Onapgo (apomorphine hydrochloride), a continuous subcutaneous infusion therapy by Supernus Pharmaceuticals for advanced Parkinson’s disease, reducing motor fluctuations and improving symptom control.
The EMR report for the Parkinson’s disease pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed Parkinson’s disease therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in Parkinson’s Disease clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for Parkinson’s disease. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of Parkinson’s disease drug candidates.
VGN‑R09b is an investigational gene therapy candidate developed by Shanghai Vitalgen BioPharma Co., Ltd. It is a recombinant adeno‑associated virus (rAAV) vector designed to deliver therapeutic genes to target pathways involved in Parkinson’s disease motor decline, aiming to improve clinical symptoms by modulating underlying disease mechanisms. In the Phase 1/2 study (NCT06480461), safety and tolerability are key endpoints, with the trial estimated to complete by July 1, 2031. The company has received U.S. Food and Drug Administration (FDA) Fast Track designation for the development of VGN-R09b.
IPS101A is an adeno‑associated virus (AAV) gene therapy candidate being investigated by Innopeutics Corporation for advanced Parkinson’s disease (Hoehn & Yahr stages 4–5). It aims to introduce genetic material into affected dopaminergic circuits to potentially improve neural function where standard therapies fail. The Phase 1 dose‑escalation trial (NCT07371338) evaluates safety, tolerability, and preliminary pharmacokinetics, with study completion expected around August 31, 2027. Innopeutics is a clinical‑stage biotech focused on innovative AAV‑based approaches for neurodegenerative conditions.
BHV‑8000 is an orally administered TYK2/JAK1 kinase inhibitor developed by Biohaven Therapeutics Ltd. It belongs to a class of small‑molecule modulators targeting neuroinflammation and immune dysregulation implicated in Parkinson’s disease progression. In a global Phase 2/3, double‑blind, placebo‑controlled study (NCT06976268), BHV‑8000 is being assessed for safety, tolerability, and efficacy in early PD, with estimated completion in September 2027. Biohaven is a clinical‑stage biopharmaceutical company advancing brain‑penetrant therapies for neurodegenerative and neurological disorders.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Parkinson’s Disease Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for Parkinson’s Disease. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into Parkinson’s disease collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
| Scope of the Report | Details |
| Drug Pipeline by Clinical Trial Phase |
|
| Route of Administration |
|
| Drug Classes |
|
| Leading Sponsors Covered |
|
| Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.