
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Recurrent malignant glioma represents one of the most aggressive and therapeutically challenging central nervous system tumors, characterized by near-universal relapse following standard treatment. Globally, high-grade gliomas, including glioblastoma, show an incidence of approximately 3-5 cases per 100,000 individuals annually, contributing to over 200,000 new cases each year. According to recurrent malignant glioma pipeline analysis by Expert Market Research, sustained clinical trial activity reflects urgent demand for novel targeted therapies and innovative biologics to address poor survival outcomes.
Major companies involved in the recurrent malignant glioma pipeline analysis include BeiGene USA, Inc., Xynomic Pharmaceuticals, Inc., and others.
Leading drugs currently in the pipeline include PCI 24781, CUE-102, and others.
The recurrent malignant glioma pipeline is driven by breakthroughs in blood-brain barrier-penetrating therapeutics, personalized neoantigen vaccines, and locoregional drug-delivery platforms such as convection-enhanced delivery and biodegradable implants. The rising molecular stratification of gliomas and expanded orphan drug incentives are accelerating precision trials and strategic biotech collaborations targeting high-mortality relapse settings.
The Recurrent Malignant Glioma Pipeline Analysis Report by Expert Market Research gives comprehensive insights into recurrent malignant glioma therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for recurrent malignant glioma. The recurrent malignant glioma report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The recurrent malignant glioma pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with recurrent malignant glioma treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to recurrent malignant glioma.
Read more about this report - Request a Free Sample
Recurrent malignant glioma remains a highly aggressive brain tumor with a poor prognosis following relapse. Outcomes after recurrence are particularly limited, as most patients have already exhausted standard first-line therapies.
Recurrent malignant glioma treatment typically includes repeat surgical resection when feasible, re-irradiation, temozolomide rechallenge, and anti-angiogenic therapy such as bevacizumab, yet durable responses are uncommon. The pipeline outlook is therefore increasingly focused on molecularly targeted and precision therapies. For instance, in August 2025, the FDA granted accelerated approval to dordaviprone (Modeyso), a first-in-class oral imipridone, for H3 K27M-mutant diffuse glioma, including recurrent disease after prior therapy, marking a significant advancement for this rare and difficult-to-treat subtype.
Recurrent malignant glioma most often follows relapse of high-grade gliomas, particularly glioblastoma, the most common and aggressive primary malignant brain tumor worldwide. Glioblastoma accounts for 45-50% of all gliomas, with a global incidence of about 2 to 5 cases per 100,000 individuals annually, translating to roughly 250,000 new cases each year. It predominantly affects older adults, with incidence rising with age, peaking in the sixth and seventh decades of life, and showing a modest male predominance. Despite standard therapy, recurrence is nearly universal, underscoring substantial unmet clinical need.
This section of the report covers the analysis of recurrent malignant glioma drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
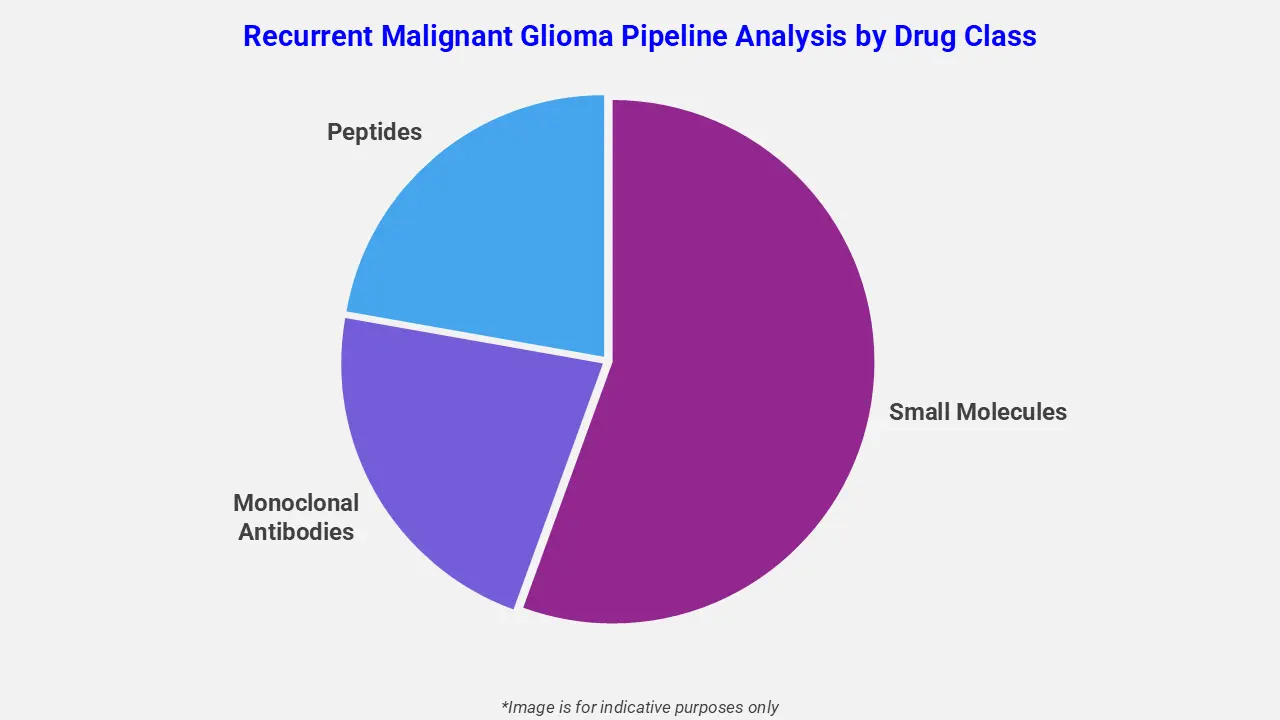
By Drug Class
The recurrent malignant glioma pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase I covers a major share of the total recurrent malignant glioma clinical trials. The report covers phase I (59%), phase II (28%), and early-phase I (11%) drugs in recurrent malignant glioma clinical trials. This distribution highlights strong early-stage research activity and a growing focus on innovative therapeutic approaches.
The drug molecule categories covered under the recurrent malignant glioma pipeline analysis include small molecules, monoclonal antibodies, and peptides. The recurrent malignant glioma report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for recurrent malignant glioma. For instance, in August 2024, the FDA approved vorasidenib (VORANIGO) as a targeted therapy for Grade 2 IDH-mutant glioma, including residual or recurrent tumors with IDH1/IDH2 mutations, improving progression-free survival and expanding precision treatment approaches in gliomas.
The EMR report for the recurrent malignant glioma pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed recurrent malignant glioma therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in recurrent malignant glioma clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for recurrent malignant glioma. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of recurrent malignant glioma drug candidates.
MVR-C5252 is an oncolytic herpes simplex virus-1 (oHSV-1) biologic developed by ImmVira, engineered to express interleukin-12 (IL-12) and an anti-PD-1 antibody for dual oncolysis and immune activation against recurrent high-grade glioma. Upon intracranial convection-enhanced delivery, it infects and lyses tumor cells while secreting IL-12 and PD-1 blockade to stimulate adaptive immunity within the tumor microenvironment. Early Phase 1 data show a favorable safety profile and immune engagement. ImmVira’s proprietary OVPENS platform underpins this novel approach, with collaboration from institutions such as Duke University driving clinical progress.
CUE-102 is a biologic Fc-fusion immunotherapy from Cue Biopharma’s Immuno-STAT® platform designed to selectively activate and expand Wilms’ Tumor 1 (WT1)-specific T cells in patients with WT1-expressing cancers, including recurrent glioblastoma. By presenting WT1 peptide and engineered interleukin-2 signals, it enhances tumor antigen-specific immune responses. Currently in Phase 1 evaluation, CUE-102 has shown encouraging tolerability and potential antitumor activity in early studies. Cue Biopharma’s immunomodulatory focus and proprietary platform aim to create precision immune therapies with reduced systemic toxicity.
PCI-24781, also known as Abexinostat, is a small-molecule pan-histone deacetylase (HDAC) inhibitor being developed by University of Nebraska in collaboration with Xynomic Pharmaceuticals. By inhibiting multiple HDAC enzyme classes, it disrupts chromatin regulation, re-expresses tumor suppressor genes, induces apoptosis, and increases reactive oxygen species, resulting in antitumor effects. While initially explored in hematologic malignancies and solid tumors, including glioma combinations, PCI-24781’s mechanism targets epigenetic modulation to halt cancer cell growth. Xynomic continues its clinical development across indications, with ongoing interest in integrating HDAC inhibition into combination regimens.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Recurrent Malignant Glioma Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for recurrent malignant glioma. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into recurrent malignant glioma collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share