
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Type 1 Diabetes continues to present a substantial global health burden, characterized by autoimmune-mediated destruction of pancreatic beta cells and lifelong insulin dependence. An estimated 9.5 million people were living with type 1 diabetes worldwide in 2025, reflecting sustained epidemiological growth and improved survival trends. As disease awareness increases and early screening improves, innovation is accelerating across immunotherapies, cell-based approaches, and advanced insulin formulations. According to Expert Market Research, the type 1 diabetes pipeline analysis highlights a strong mid-stage clinical landscape, signaling steady therapeutic evolution and long-term market potential.
Major companies involved in type 1 diabetes pipeline analysis include Creative Medical Technology Holdings Inc, PolTREG S.A., and others.
Leading drugs currently in the pipeline include GNTI-122, CNP-103, and others.
The type 1 diabetes pipeline is propelled by disease-modifying strategies, including antigen-specific immunotherapies and regulatory T-cell platforms targeting autoimmune destruction at earlier stages. Screening expansion for presymptomatic populations, biomarker-driven staging, and combination approaches pairing immune modulation with beta-cell preservation are unlocking preventive treatment pathways beyond lifelong insulin dependence.
The Type 1 Diabetes Pipeline Analysis Report by Expert Market Research gives comprehensive insights into type 1 diabetes therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for type 1 diabetes. The type 1 diabetes report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The type 1 diabetes pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with type 1 diabetes treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to type 1 diabetes.
Read more about this report - Request a Free Sample
The type 1 diabetes pipeline outlook reflects a shift beyond lifelong insulin replacement toward disease-modifying and more accessible therapeutic options.
Type 1 diabetes treatment relies on intensive insulin therapy, including rapid-acting and long-acting insulin analogs, supported by continuous glucose monitoring and insulin pump technologies to maintain glycemic control. For instance, in February 2025, the U.S. Food and Drug Administration approved Merilog (insulin-aspart-szjj), the first rapid-acting insulin biosimilar to improve glycemic control in adults and pediatric patients with diabetes mellitus. This approval represents a significant step in expanding more affordable insulin options while next-generation immunotherapies and beta-cell–preserving strategies continue advancing in the pipeline.
Type 1 diabetes remains a significant and growing global health burden, with an estimated 9.5 million people living with the disease worldwide in 2025. The epidemiology reflects improved survival rates, population growth, and increasing recognition across regions. A substantial proportion of affected individuals are aged 20-59 years, while children and adolescents continue to represent a meaningful share of cases. Notably, over one-fifth of people with type 1 diabetes reside in low- and middle-income countries, highlighting disparities in diagnosis and care access that continue to shape global disease outcomes.
This section of the report covers the analysis of type 1 diabetes drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
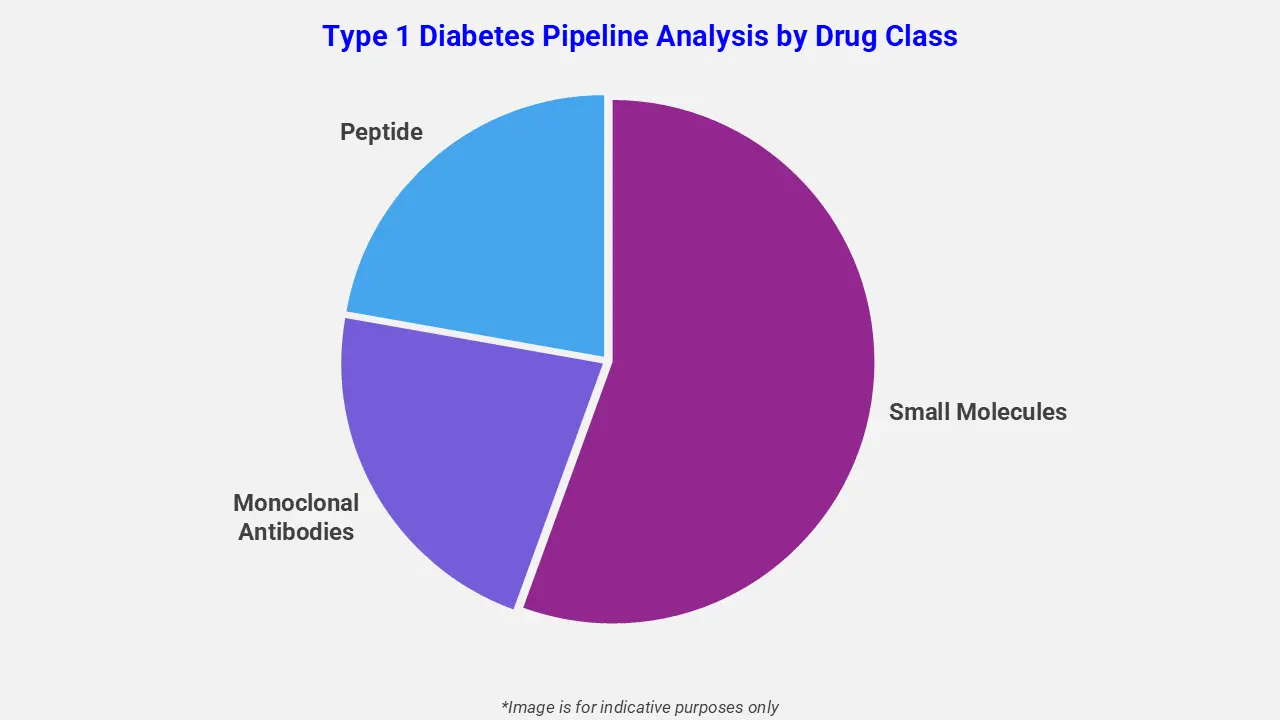
By Drug Class
The type 1 diabetes pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total type 1 diabetes clinical trials. The report covers Phase I (28%), Phase II (43%), Phase III (14%), Phase IV (9%), and Early Phase I (6%) drugs. These findings highlight that mid-stage development dominates the type 1 diabetes pipeline, reflecting active clinical exploration and the potential for future therapeutic advancements.
The drug molecule categories covered under the type 1 diabetes pipeline analysis include monoclonal antibodies, small molecules, and peptides. The type 1 diabetes report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for type 1 diabetes. In June 2023, the U.S. Food and Drug Administration approved Lantidra (donislecel), the first allogeneic pancreatic islet cell therapy for adults with type 1 diabetes and recurrent severe hypoglycemia. This therapy uses donor islet cells to produce insulin, offering a novel therapeutic option beyond traditional management for patients struggling with severe low blood sugar.
The EMR report for the type 1 diabetes pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed type 1 diabetes therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in type 1 diabetes clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for type 1 diabetes. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of type 1 diabetes drug candidates.
SAR442970 is an investigational anti-CD40L monoclonal antibody developed by Sanofi that targets both tumor necrosis factor-α (TNF-α) and OX40L to modulate adaptive immune responses. By simultaneously inhibiting these inflammatory pathways, it aims to reduce immune-mediated tissue damage and preserve organ function. Currently in Phase 2 trials across autoimmune indications, including type 1 diabetes and Crohn’s disease, Sanofi’s broader immunology portfolio leverages SAR442970’s unique dual-target approach to expand beyond single-pathway biologics in chronic inflammatory diseases.
RGB-5088 is a regenerative islet cell therapy developed by Hangzhou Reprogenix Bioscience, Inc. that uses chemically induced pluripotent stem cell (CiPSC)-derived pancreatic islet cells to restore endocrine function in type 1 diabetes. In early Phase 1 trials, these transplanted islet cells aim to replace dysfunctional insulin-producing cells and normalize glucose regulation. Reprogenix leverages its stem cell and reprogramming expertise to advance RGB-5088, positioning it as a pioneering cellular approach that could reduce or eliminate dependence on exogenous insulin if successful.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Type 1 Diabetes Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for type 1 diabetes. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into type 1 diabetes collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.