
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Endometrial cancer is a malignancy that originates in the lining of the uterus (endometrium), most commonly affecting postmenopausal women due to hormonal and metabolic risk factors. According to research by Songkun Gao et al., 2025, the disease accounts for more than 2% of global cancer-related mortality, highlighting its significant clinical burden. Current therapeutic approaches include surgery, radiation, chemotherapy, hormonal therapy, and targeted immunotherapies such as checkpoint inhibitors. According to the endometrial cancer pipeline analysis by Expert Market Research, increasing biomarker-driven drug development, novel combination regimes, and precision oncology strategies are expected to drive robust pipeline expansion and treatment advancements in the coming years.
Major companies involved in the endometrial cancer pipeline analysis include BioNTech SE, and others.
Leading drugs currently in the pipeline include BNT323/DB-1303, HS-20089, BL-M07D1, and others.
The robust pipeline growth in endometrial cancer is driven by immunotherapy combinations, biomarker-guided targeted agents, and expanding indications across recurrent, advanced, and molecularly defined patient subgroups.
The Endometrial Cancer Pipeline Analysis Report by Expert Market Research gives comprehensive insights into endometrial cancer therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for endometrial cancer. The endometrial cancer report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The endometrial cancer pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with endometrial cancer treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to endometrial cancer.
Read more about this report - Request a Free Sample
Endometrial cancer is a malignancy originating in the endometrium, the inner lining of the uterus. It typically develops due to prolonged estrogen exposure unopposed by progesterone, genetic mutations, or metabolic risk factors such as obesity. Abnormal cellular proliferation leads to tumor formation, commonly presenting with postmenopausal bleeding and pelvic discomfort.
Endometrial cancer treatment involves surgery, primarily hysterectomy with bilateral salpingo-oophorectomy, followed by risk-adapted radiotherapy or chemotherapy. Advanced or recurrent disease may require targeted therapy, immunotherapy, or hormone therapy based on molecular profiling and disease stage. In June 2024, the U.S. Food and Drug Administration approved pembrolizumab with carboplatin and paclitaxel for primary advanced or recurrent endometrial carcinoma, followed by maintenance pembrolizumab. Developed by Merck, the regimen demonstrated significant progression-free survival improvement in mismatch repair–deficient and proficient patient cohorts in the KEYNOTE-868 trial.
The pipeline has strong growth potential driven by rising disease incidence and evolving therapeutic strategies. According to Songkun Gao et al., 2025, incidence among postmenopausal women aged 55 years and above is projected to increase by 6.5% by 2036, despite an 8.0% decline in mortality. As per Jason D. Wright et al., 2025, uterine cancer incidence in the United States may reach 74.2 per 100,000 in White women and 86.9 per 100,000 in Black women by 2050. Additionally, according to Health Data Insight, the United Kingdom reports over 9,700 new cases annually, supporting continued pipeline innovation and targeted drug development demand.
This section of the report covers the analysis of endometrial cancer drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
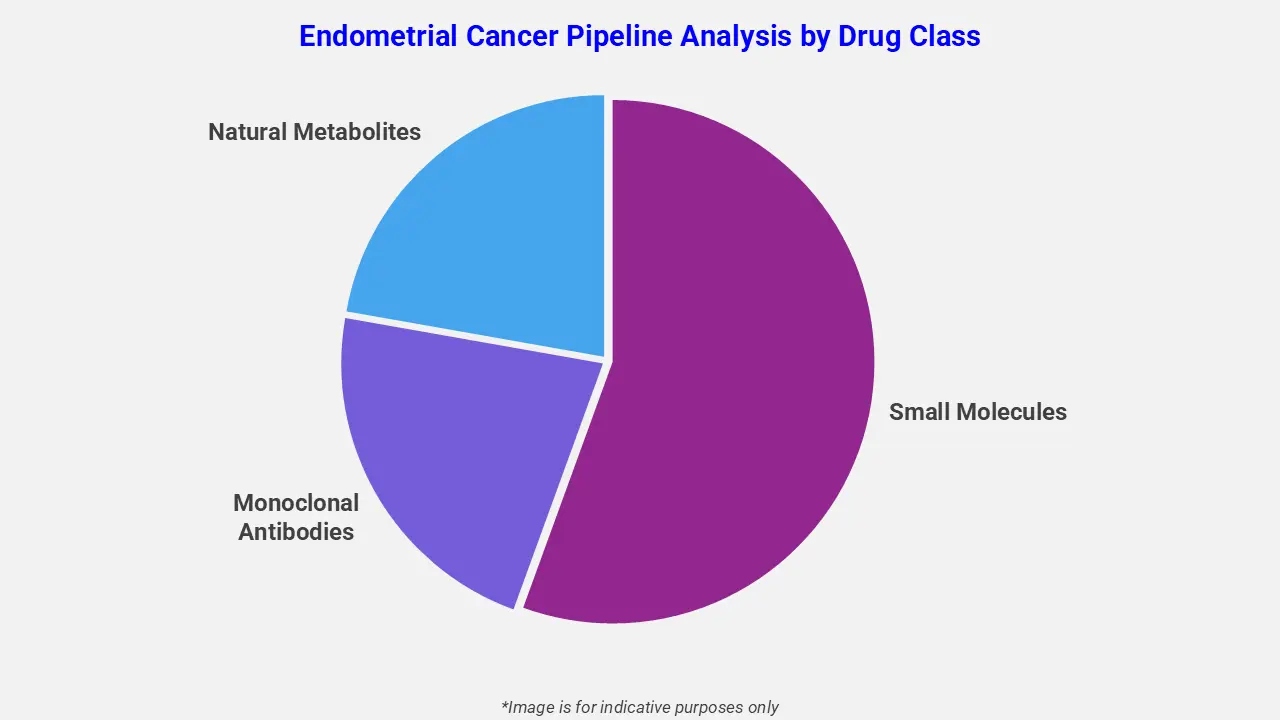
By Drug Class
The endometrial cancer pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with 49%, covers a major share of the total endometrial cancer clinical trials, reflecting strong mid-stage clinical advancement and commercialization potential. Phase I at 37% and phase III at 11% indicate sustained innovation and late-stage progression. This balanced progression supports therapeutic advancements, regulatory approvals, and overall market expansion.
The drug molecule categories covered under the endometrial cancer pipeline analysis include small molecules, monoclonal antibodies, and natural metabolites. The endometrial cancer report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for endometrial cancer. HER2-targeted antibody–drug conjugates are emerging as promising options for endometrial cancer management. For instance, trastuzumab deruxtecan is being investigated in the Phase 3 DESTINY-Endometrial01 trial in combination with immune checkpoint inhibitors such as pembrolizumab or rilvegostomig. It selectively binds to HER2-expressing tumor cells and delivers a cytotoxic topoisomerase I inhibitor payload, thereby enhancing antitumor activity and improving therapeutic outcomes.
The EMR report for the endometrial cancer pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed endometrial cancer therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in endometrial cancer clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for endometrial cancer. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of endometrial cancer drug candidates.
BNT323/DB-1303 is a third-generation HER2-targeting antibody–drug conjugate sponsored by BioNTech SE. This Phase III trial is evaluating its anti-tumor efficacy and safety in previously treated, HER2-expressing recurrent endometrial cancer. The study is comparing BNT323 with investigator’s choice chemotherapy in Cohort 1 by examining progression-free survival, while Cohort 2 is assessing objective response rate with BNT323 monotherapy. Built on DualityBio’s DITAC platform, the drug is delivering a topoisomerase-I inhibitor payload directly to HER2-positive tumor cells, potentially enhancing cytotoxic activity while maintaining a manageable safety profile, alongside pharmacokinetics and quality-of-life assessments.
HS-20089 is an investigational B7-H4–targeted antibody–drug conjugate sponsored by Jiangsu Hansoh Pharmaceutical Group Co., Ltd, and the Phase 2 study is evaluating its efficacy, safety, pharmacokinetics, and immunogenicity as monotherapy in patients with recurrent or metastatic endometrial cancer who have progressed after platinum-based chemotherapy. The drug comprises a humanized IgG1 anti-B7-H4 monoclonal antibody linked to a topoisomerase I inhibitor payload via a protease-cleavable linker, enabling targeted cytotoxic delivery to tumor cells. It is being administered intravenously at weight-based doses, while global development activities are also being supported through licensing collaboration with GlaxoSmithKline.
BL-M07D1 is being investigated in a Phase 1 open-label study sponsored by SystImmune Inc. to evaluate its safety, tolerability, pharmacokinetics, and preliminary efficacy in patients with HER2-expressing advanced solid tumors, including endometrial cancer. The objective of this phase is to determine optimal dosing through dose escalation, dose finding, and expansion cohorts. BL-M07D1 is a HER2-targeted antibody–drug conjugate based on trastuzumab with dual HER2-binding domains, enabling receptor internalization and lysosomal payload release that induces genotoxic stress and tumor cell death. The drug is being administered on Day 1 of a continuous 21-day treatment cycle across multiple global centers.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Endometrial Cancer Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for endometrial cancer. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into endometrial cancer collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share