
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Lewy body disease (LBD) is a progressive neurodegenerative disorder characterized by abnormal alpha-synuclein aggregation, leading to cognitive decline in Lewy body dementia, hallucinations, and Parkinsonian symptoms. According to Sonja W. Scholz et al., 2025, LBD represents one of the most common dementias among individuals aged ≥65. Current therapeutic approaches include cholinesterase inhibitors for dementia, antipsychotics, dopaminergic agents, and emerging disease-modifying therapies for Lewy body disease targeting alpha-synuclein pathology. According to the Lewy body disease pipeline analysis by Expert Market Research, growing focus on biomarker-based diagnosis in neurodegenerative diseases, precision medicine in dementia care, and neuroprotective drug development, along with rising aging populations and unmet clinical needs, is expected to drive pipeline expansion and market growth in the coming years.
Major companies involved in the Lewy body disease pipeline analysis include ACADIA Pharmaceuticals Inc., EIP Pharma Inc., and others.
Leading drugs currently in the pipeline include ACP-204, Neflamapimod, hUC-MSC-sEV-001 nasal drops, and others.
The pipeline expansion is driven by alpha-synuclein-targeting therapies, neuroinflammation modulators in dementia, and drug repurposing in neurodegenerative diseases, alongside biomarker-guided trials and improved diagnostics, accelerating candidate progression and attracting partnerships and renewed investment globally.
The Lewy Body Disease Pipeline Analysis Report by Expert Market Research gives comprehensive insights into Lewy body disease therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for Lewy body disease. The Lewy body disease report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The Lewy body disease pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with Lewy body disease treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to Lewy body disease.
Read more about this report - Request a Free Sample
Lewy body dementia (LBD) is a complex neurodegenerative brain disorder driven by the accumulation of alpha-synuclein protein deposits, commonly referred to as Lewy bodies. These abnormal deposits interfere with neuronal signaling and brain function, resulting in a spectrum of Lewy body dementia symptoms such as progressive cognitive impairment, persistent visual hallucinations, parkinsonian motor symptoms, and REM sleep behavior disorder. Patients often experience marked fluctuations in attention and alertness, making clinical diagnosis challenging, especially due to its overlap with Parkinson’s disease dementia and Alzheimer’s disease pathology.
Lewy body disease treatments focus on managing symptoms, including psychosis, cognitive impairment, and motor dysfunction. Approved and off-label medications, such as pimavanserin, quetiapine, clozapine, olanzapine, donepezil, rivastigmine, and galantamine, are used to reduce hallucinations, delusions, and cognitive decline. Pimavanserin, in the lewy body disease pipeline, is undergoing targeted use for psychosis in Parkinson’s disease dementia. Data from the Phase 3 Harmony trial, published in March 2024, show a 95% reduced relapse risk in patients, highlighting its efficacy and tolerability without worsening motor or cognitive functions.
The pipeline is gaining significant attention due to the rising prevalence of dementia with Lewy bodies (DLB). According to Sonja W. Scholz et al., 2025, Lewy body dementia is the most prevalent neurodegenerative form of dementia in individuals aged 65 and older. As per Carolyn W. Zhu et al., 2024, DLB involves the deposition of alpha-synuclein protein in brain neurons and represents the second most common degenerative dementia after Alzheimer’s disease, accounting for up to 20% of dementia cases in the United States. According to Akiyo Yumoto, et al., 2024, Epidemiologically, approximately 800,000 people in Japan are affected by DLB. This underscores the targeted therapies for Lewy body dementia and improved diagnostic strategies.
This section of the report covers the analysis of Lewy body disease drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
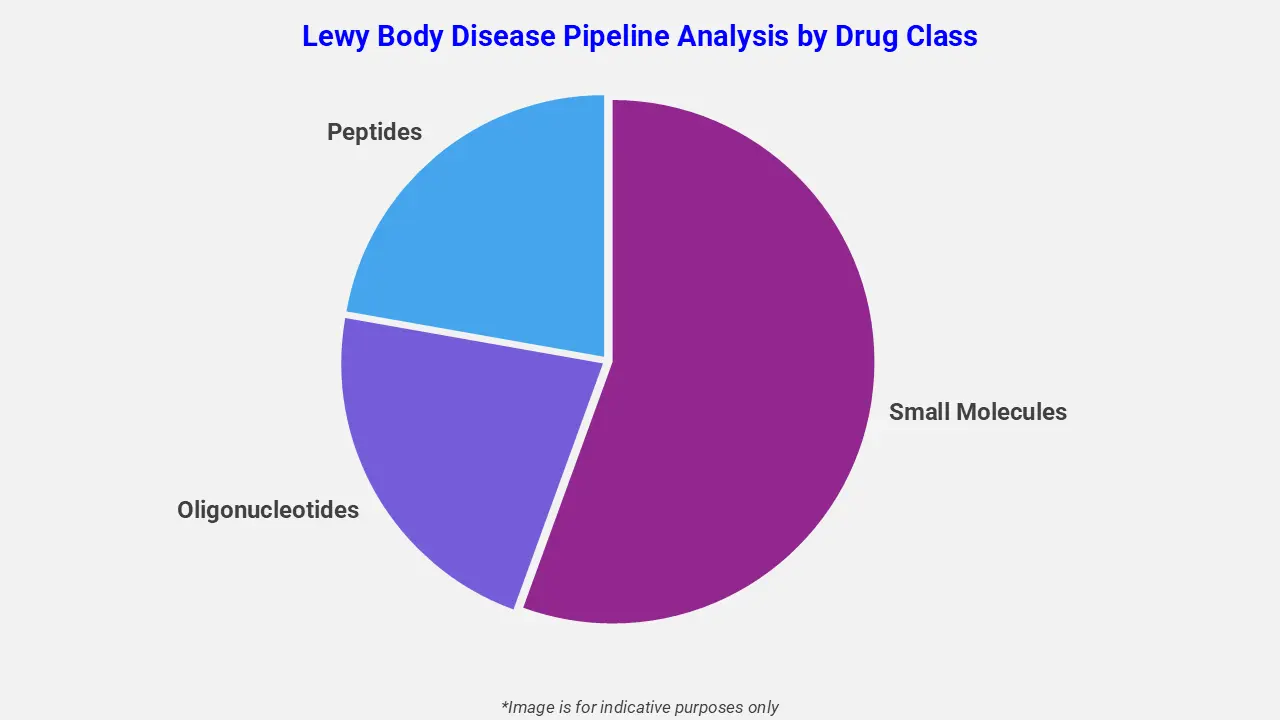
By Drug Class
The Lewy body disease pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with 43%, covers a major share of the total Lewy body disease clinical trials, indicating strong research momentum. Phase IV accounts for 21%, supported by post-marketing surveillance and real-world evidence gathering, enhancing long-term treatment validation. Phase I represents 18%, fueled by safety assessments and dose optimization studies. Phase III holds 11%, reflecting pivotal trials and regulatory preparation. Early phase I contributes around 7%, focusing on initial safety and pharmacokinetics evaluations.
The drug molecule categories covered under the Lewy body disease pipeline analysis include small molecules, oligonucleotides, and peptides. The Lewy body disease report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for Lewy body disease. Neuromodulatory therapies for dementia are gaining attention in the Lewy body disease pipeline to address cognitive and motor impairments. For example, Zervimesine, an investigational oral medication, is under evaluation in an expanded access program. It targets abnormal alpha-synuclein protein deposits in the brain and has shown potential in slowing the progression of dementia with Lewy body symptoms, offering a promising treatment approach for patients.
The EMR report for the Lewy body disease pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed Lewy body disease therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in Lewy body disease clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for Lewy body disease. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of Lewy body disease drug candidates.
ACP-204, an orally administered inverse agonist, is being developed by ACADIA Pharmaceuticals Inc. for the treatment of Lewy body dementia psychosis (LBDP). This Phase 3, open-label extension study is enrolled by invitation and is designed to examine the long-term safety, tolerability, and sustained efficacy of ACP-204 in adults with LBDP. The drug works by selectively targeting 5-HT2A receptors in the brain, modulating neurotransmitter activity to reduce psychotic symptoms without affecting motor function. Building on insights from pimavanserin, ACP-204 is continuing to be evaluated for its potential to provide meaningful symptom relief over 52 weeks and strengthen therapeutic options for Lewy body dementia.
Neflamapimod, sponsored by CervoMed, is a small-molecule oral drug targeting the p38ɑ kinase protein, a key driver of inflammation and neuronal cell death in dementia with Lewy bodies (DLB). Inhibiting this pathway prevents synaptic dysfunction, improving communication between brain cells, and slowing disease progression. The upcoming Phase 3 study is examining the drug’s efficacy and safety in approximately 300 participants over 24 weeks, focusing on individuals with low Alzheimer’s biomarkers. This trial evaluates cognitive and functional outcomes to determine if neflamapimod can become the first disease-modifying drug for Lewy body dementia.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Lewy Body Disease Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for Lewy body disease. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into Lewy body disease collaborations, regulatory environments, and potential growth opportunities.
Parkinson’s Disease Drugs Market




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|---|---|
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share