
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Preeclampsia is a serious hypertensive disorder of pregnancy characterized by elevated blood pressure and organ dysfunction, posing significant risks to both the mother and fetus. Globally, it affects approximately 2-8% of pregnancies, contributing substantially to maternal and neonatal morbidity and mortality. Despite its prevalence, treatment remains largely supportive, with delivery as the only definitive solution. According to Expert Market Research, the preeclampsia pipeline analysis highlights increasing investment in targeted therapies addressing angiogenic imbalance and placental dysfunction, signaling a shift toward disease-modifying strategies in maternal-fetal medicine.
Major companies involved in the preeclampsia pipeline analysis include Comanche Biopharma, DiaMedica Therapeutics Inc., and others.
Leading drugs currently in the pipeline include 17-OHPC, CBP-4888, and others.
A key market driver in the preeclampsia pipeline is the shift toward disease-modifying therapies targeting placental angiogenic imbalance, particularly sFlt-1 and endothelin pathways. Advancements in RNA-based therapeutics and biomarker-guided patient stratification are accelerating precision approaches, enabling earlier intervention and extending pregnancy duration, an unmet clinical need driving focused innovation and investment.
The Preeclampsia Pipeline Analysis Report by Expert Market Research gives comprehensive insights into preeclampsia therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for preeclampsia. The preeclampsia report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The preeclampsia pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with preeclampsia treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to preeclampsia.
Read more about this report - Request a Free Sample
The preeclampsia pipeline outlook reflects growing momentum to address a condition historically managed with limited therapeutic options. Standard care primarily involves blood pressure control, seizure prophylaxis with magnesium sulfate, and timely delivery, which remains the only definitive treatment. However, innovation is expanding beyond symptomatic management. In February 2025, the U.S. Food and Drug Administration granted 510(k) clearance to Roche’s Elecsys sFlt-1/PlGF ratio test for preeclampsia risk stratification, enabling improved prediction of severe disease progression. This regulatory milestone strengthens biomarker-driven approaches and supports precision-based intervention strategies in maternal-fetal medicine.
Preeclampsia is a hypertensive disorder of pregnancy affecting an estimated about 4.4% of pregnancies worldwide, with regional variability. Low-income regions often report higher rates than high-income countries. Overall global prevalence estimates range roughly 2-8% of pregnancies, contributing significantly to maternal and perinatal morbidity and mortality. Risk factors include advanced maternal age, obesity, and chronic hypertension, and disparities persist between regions with differing healthcare access and prenatal care quality.
This section of the report covers the analysis of preeclampsia drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
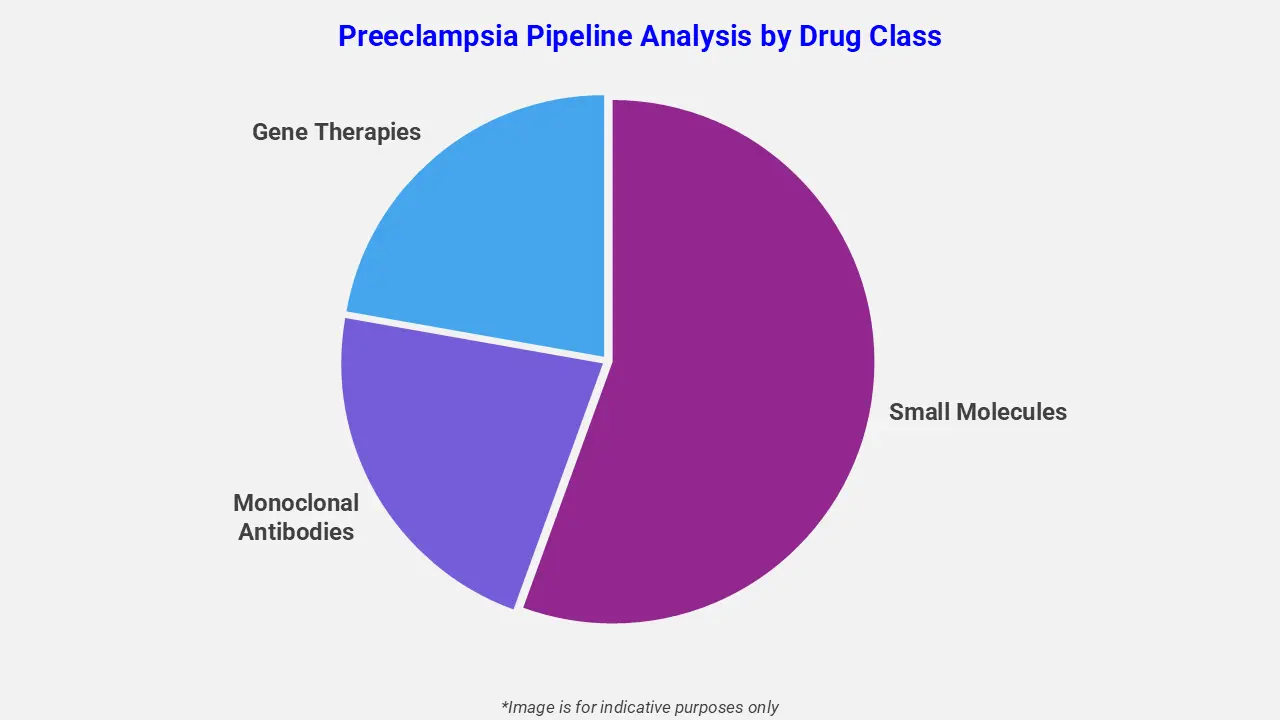
By Drug Class
The preeclampsia pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total preeclampsia clinical trials. Phase II and Phase IV each account for 26%, followed by Phase III (20%), Phase I (15%), and Early Phase I (13%). This distribution highlights a strong focus on mid- to late-stage clinical development, reflecting increasing momentum toward advancing potential therapeutic options for preeclampsia management.
The drug molecule categories covered under the preeclampsia pipeline analysis include small molecules, monoclonal antibodies, and gene therapies. The preeclampsia report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for preeclampsia. In December 2025, DiaMedica Therapeutics Inc. announced completion of an in-person pre-IND meeting with the U.S. FDA regarding its planned DM199 preeclampsia study, with an additional non-clinical study requested and results expected by Q2 2026. Interim Phase 2 data show safety, reduced blood pressure, and uterine artery dilation with no evidence of placental transfer, supporting ongoing development of DM199 for preeclampsia.
The EMR report for the preeclampsia pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed preeclampsia therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in preeclampsia clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for preeclampsia. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of preeclampsia drug candidates.
CBP-4888 is an investigational siRNA (small interfering RNA) therapeutic developed by Comanche Biopharma to treat preeclampsia by targeting the root molecular cause of the disease. It consists of a fixed-dose combination of two lipid-conjugated siRNAs designed to downregulate placental soluble fms-like tyrosine kinase-1 (sFlt1), a key pathogenic mediator believed to drive endothelial dysfunction, hypertension, and disease progression. By reducing sFlt1 production, CBP-4888 aims to alleviate maternal symptoms and potentially extend pregnancy safely. Comanche Biopharma, a clinical-stage maternal health biotech, is advancing the candidate through early clinical trials while securing regulatory designations such as Fast Track, orphan, and Innovation Passport status to accelerate development.
Metformin is an oral biguanide antidiabetic agent widely used for type 2 diabetes that is being investigated for preeclampsia prevention and treatment due to its effects on metabolic, inflammatory, and endothelial pathways. It activates AMP-activated protein kinase (AMPK) and may inhibit inflammatory and oxidative stress signaling (NF-κB/sFlt-1), which are implicated in preeclampsia pathogenesis, suggesting a potential benefit in improving placental function. Clinical studies and trials continue to evaluate metformin’s ability to reduce preeclampsia incidence or severity, with ongoing research supported by obstetric and maternal-fetal medicine investigators aiming to repurpose this established therapy for high-risk pregnancies.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Preeclampsia Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for preeclampsia. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into preeclampsia collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share