
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Renal insufficiency is a medical condition characterized by a decline in kidney function, commonly identified by an estimated glomerular filtration rate (eGFR) below 60 mL/min/1.73 m², which limits the kidneys’ ability to filter waste and maintain fluid and electrolyte balance. According to Theresia A. Ottaru et al., 2024, the global prevalence of renal insufficiency among adults living with HIV is reported to be approximately 3% to 7%. According to the renal insufficiency pipeline analysis by Expert Market Research, the therapeutic pipeline includes renin–angiotensin system inhibitors, SGLT2 inhibitors, anti-inflammatory therapies, and emerging nephroprotective agents. Increasing emphasis on early detection, targeted therapies, and advancements in nephrology research is expected to support steady pipeline expansion and drive growth in renal insufficiency drug development in the coming years.
Major companies involved in the renal insufficiency pipeline analysis include Daewoong Pharmaceutical Co. Ltd., Eli Lilly and Company, and others.
Leading drugs currently in the pipeline include DWP16001, LY3537982, LY3841136, and others.
Strong pipeline expansion is driven by novel anti-fibrotic and anti-inflammatory candidates, increasing focus on disease-modifying therapies, and rising clinical trials evaluating targeted biologics and small molecules to slow kidney function decline.
The Renal Insufficiency Pipeline Analysis Report by Expert Market Research gives comprehensive insights into renal insufficiency therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for renal insufficiency. The renal insufficiency report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The renal insufficiency pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with renal insufficiency treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to renal insufficiency.
Read more about this report - Request a Free Sample
Renal insufficiency is a condition where the ability of the kidneys to filter waste and excess fluids from the blood is reduced. It can occur due to diabetes, hypertension, glomerulonephritis, or prolonged kidney injury, leading to toxin accumulation, fluid imbalance, and complications such as anaemia, electrolyte disturbances, and cardiovascular issues. Early detection and management are essential to prevent progression.
Renal insufficiency treatment focuses on managing underlying causes, controlling blood pressure, and correcting complications. Therapeutic approaches include erythropoiesis-stimulating agents, iron supplementation, lifestyle modifications, dialysis, and innovative oral medications. Jesduvroq (daprodustat) exemplifies progress in the market, offering a treatment option for adults on dialysis with anemia a first-in-class oral HIF-PHI therapy. Approved by the US FDA in February 2023, Jesduvroq stabilizes hypoxia-inducible factors, promotes erythropoiesis, and provides a convenient, effective treatment option.
The pipeline is expanding in response to the growing global burden of kidney disease. According to Aminu K. Bello et al., 2025, the median global prevalence of chronic kidney disease is 9.5%, with the highest rates in Eastern and Central Europe (12.8%). Chronic hemodialysis is available in 98% of countries, peritoneal dialysis in 79%, and kidney transplantation in 70%. As per Theresia A. Ottaru et al., 2024, renal insufficiency among adults living with HIV ranges from 3–7% globally, reaching 12% in Sub-Saharan Africa. Limited access to kidney replacement therapies and workforce disparities underscore the need for targeted pipeline development.
This section of the report covers the analysis of renal insufficiency drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
By Drug Class
The renal insufficiency pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II, with 33%, covers a major share of the total renal insufficiency clinical trials, reflecting strong clinical development momentum. Phase IV contributes 22%, and phase III holds 21%, indicating advanced therapies approaching commercialization. Phase I represents 20%, supporting early clinical validation and demonstrating ongoing exploratory research. This diverse pipeline has the potential to significantly enhance treatment options and market growth.
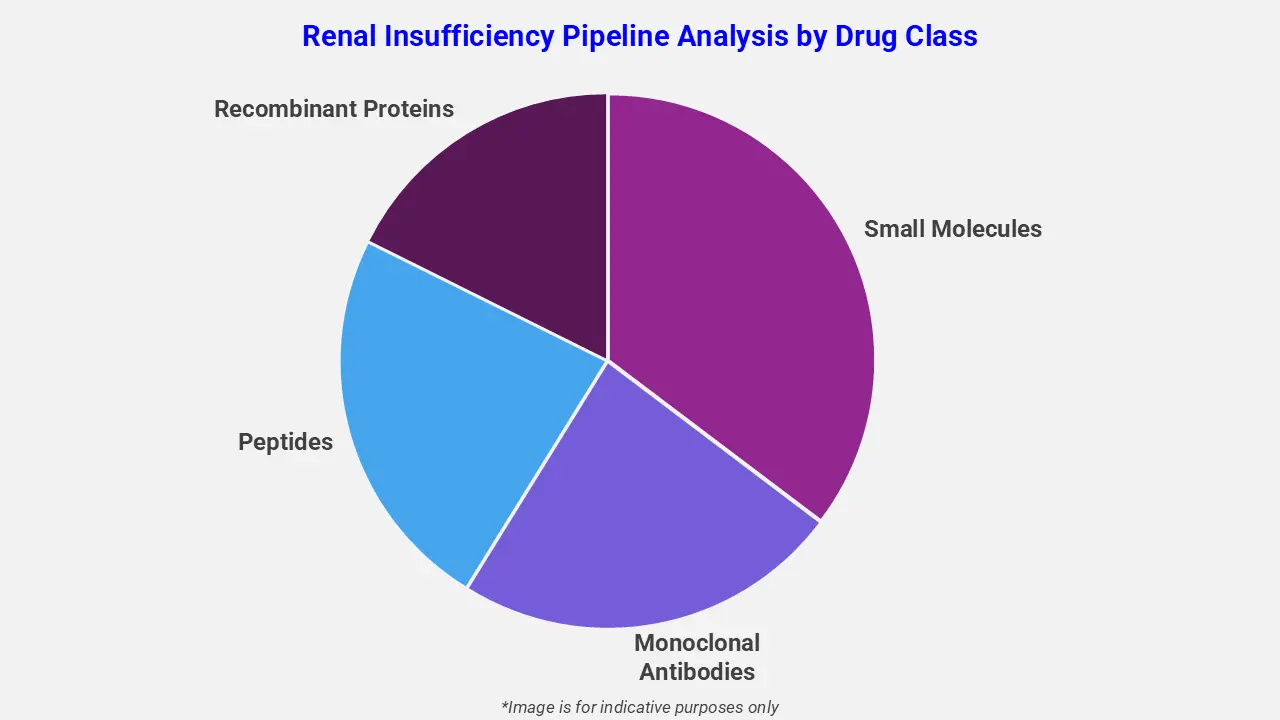
The drug molecule categories covered under the renal insufficiency pipeline analysis include small molecules, monoclonal antibodies, peptides, and recombinant proteins. The renal insufficiency report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for renal insufficiency. Enzyme replacement therapies are emerging as promising interventions in the renal insufficiency drug pipeline. For example, ilofotase alfa, a recombinant alkaline phosphatase, is under Phase 2 evaluation for the prevention of cardiac surgery-associated renal damage. It works by dephosphorylating pro-inflammatory molecules and generating tissue-protective adenosine, thereby reducing acute kidney injury and improving post-surgical renal outcomes.
The EMR report for the renal insufficiency pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed renal insufficiency therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in renal insufficiency clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for renal insufficiency. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of renal insufficiency drug candidates.
Enavogliflozin (DWP16001) is an investigational, highly selective sodium–glucose co-transporter-2 (SGLT-2) inhibitor being developed by Daewoong Pharmaceutical Co., Ltd. for patients with type 2 diabetic nephropathy and moderate renal impairment (CKD stage 3). The ongoing Phase 3 randomized, double-blind, placebo-controlled, multicenter trial is evaluating the efficacy and safety of once-daily oral enavogliflozin 0.3 mg by assessing its ability to improve renal and metabolic outcomes through insulin-independent urinary glucose excretion. The study is enrolling approximately 348 participants and is expected to begin in September 2024, with primary completion anticipated in September 2026 and final study completion projected for January 2027.
Olomorasib (LY3537982) is being evaluated in a Phase 1 clinical study sponsored by Eli Lilly and Company to investigate its pharmacokinetics following a single oral dose in participants with renal insufficiency compared with those having normal kidney function. Olomorasib is an investigational, oral, highly selective second-generation KRAS G12C inhibitor designed to block the KRAS G12C mutant protein, thereby suppressing abnormal cell signaling pathways involved in disease progression. The study is examining drug exposure, metabolism, and appropriate dosing in patients with varying degrees of renal impairment. The trial is recruiting participants and is expected to be completed in March 2026.
Eloralintide (LY3841136) is being evaluated in a Phase 1 clinical study sponsored by Eli Lilly and Company to assess its pharmacokinetics, safety, and systemic exposure in participants with renal insufficiency compared with individuals with normal renal function. The study examines how the body absorbs, distributes, and eliminates the drug after a single subcutaneous administration. Eloralintide is a synthetic amylin analog peptide designed to selectively activate amylin receptors, potentially improving metabolic regulation and with a reduced risk of immunogenicity risk. The multicenter, open-label study started in February 2026 and is expected to reach completion in the October 2026.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Renal Insufficiency Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for renal insufficiency. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into renal insufficiency collaborations, regulatory environments, and potential growth opportunities.
Acute Kidney Injury Treatment Market
Acute Kidney Injury Drug Pipeline Analysis Report
Acute Renal Failure/Acute Kidney Injury Epidemiology Forecast




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share