
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
Tuberculosis (TB) is a contagious airborne bacterial infection caused by Mycobacterium tuberculosis, primarily affecting the lungs but potentially impacting other organs. According to the World Health Organization, around 10.7 million people developed TB globally in 2024, reflecting a significant disease burden. Current therapies include first-line antibiotics, multidrug-resistant TB regimens, and emerging all-oral treatments. According to the tuberculosis pipeline analysis by expert market research, there is growing focus on novel drug combinations, shorter regimens, and host-directed therapies. Increasing drug resistance, rising R&D investments, and advancements in diagnostics are expected to drive pipeline expansion and market growth in the coming years.
Major companies involved in tuberculosis pipeline analysis include BioNTech SE, Quratis Inc., and others.
Leading drugs currently in the pipeline include JDB0131, BNT164a1, QTP101, and others.
Robust clinical activity driven by novel drug classes, shorter treatment regimens, and rising investment in multidrug-resistant TB therapies is expected to accelerate approvals and expand therapeutic options in coming years.
The Tuberculosis Pipeline Analysis Report by Expert Market Research gives comprehensive insights into tuberculosis therapeutics currently undergoing clinical trials. It covers various aspects related to the details of each of these drugs under development for tuberculosis. The tuberculosis report assessment includes the analysis of over 100 pipeline drugs and 50+ companies. The tuberculosis pipeline landscape will include an analysis based on efficacy and safety measure outcomes published for the trials, including their adverse effects on patients suffering from the condition, and alignment with tuberculosis treatment guidelines to ensure optimal care practices.
The assessment part will include a detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration, and ongoing product development activities related to tuberculosis.
Read more about this report - REQUEST FREE SAMPLE COPY IN PDF
Tuberculosis (TB) is an infectious disease caused by the bacterium Mycobacterium tuberculosis. It primarily affects the lungs but can spread to other organs. Transmission occurs via airborne droplets from coughs or sneezes of infected individuals, leading to latent or active infection when bacteria invade lung tissue and multiply.
Tuberculosis treatments involve standard regimens like HRZE (isoniazid, rifampin, pyrazinamide, ethambutol) for drug-sensitive TB, typically lasting 6 months. Drug-resistant cases use longer courses with bedaquiline, pretomanid, and linezolid, emphasizing adherence to prevent resistance. In November 2025, TB Alliance's Phase 2 NC-009 trial results revealed that sorfequiline (TBAJ-876), combined with pretomanid and linezolid in the SPaL regimen, outperformed standard HRZE for drug-sensitive TB, showing potential to shorten treatment duration while maintaining a comparable safety profile.
Tuberculosis remains a pressing global health challenge, demanding robust drug pipeline advancements. According to the World Health Organization (WHO) Global Tuberculosis Report 2025, an estimated 10.7 million people (95% uncertainty interval: 9.9–11.5 million) developed TB in 2024, equating to 131 incident cases per 100,000 population. Men comprised 54% of cases, women 35%, and children/young adolescents 11%. In India, per Ghanshyam Ahirwar et al. (2025) citing India TB Report 2024, the country bore 26% of the global burden with 2.7 million cases in 2023, achieving 89% treatment coverage and a 17.7% incidence decline to 195 per 100,000 population. Strategic drug innovations are vital for accelerating TB elimination.
This section of the report covers the analysis of tuberculosis drug candidates based on several segmentations, including:
By Phase
The pipeline assessment report covers 50+ drug analyses based on phase:
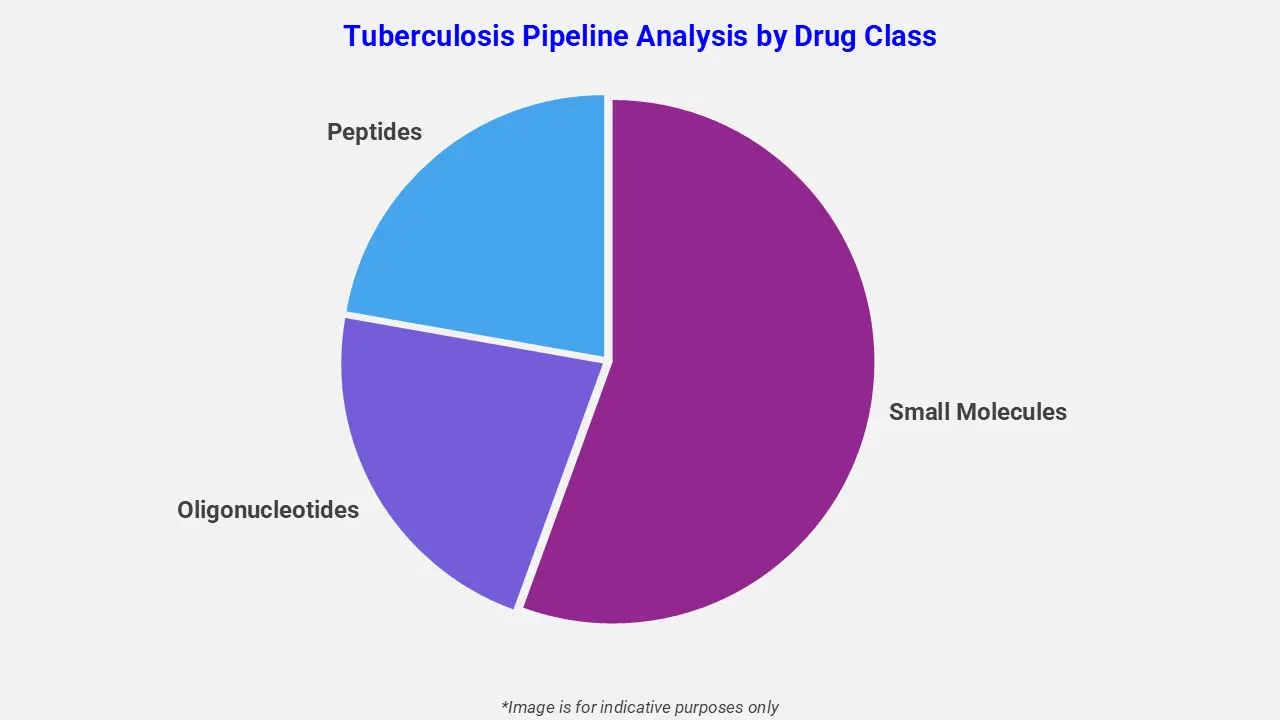
By Drug Class
The tuberculosis pipeline analysis report covers 50+ drug analyses based on drug classes:
By Route of Administration
The pipeline assessment report covers 50+ drug analyses based on the route of administration:
The report covers phase I, phase II, phase III, phase IV, and early-phase drugs. The coverage includes an in-depth analysis of each drug across these phases. According to EMR analysis, phase II covers a major share of the total tuberculosis clinical trials, commands 38%, accelerating efficacy validation. Phase III with 35%, ensuring regulatory readiness. Phase I contributes 15%, fostering novel discoveries. Phase IV adds 12%, enhancing post-approval optimization. Overall, this phase-wise distribution reflects a well-balanced and advancing pipeline, indicating sustained innovation and a strong trajectory toward improved tuberculosis treatment outcomes.
The drug molecule categories covered under the tuberculosis pipeline analysis include small molecules, oligonucleotides, and peptides. The tuberculosis report provides a comparative analysis of the drug classes for each drug in various phases of clinical trials for tuberculosis. Novel drug classes for tuberculosis are advancing to address drug-resistant strains and improve treatment outcomes. For instance, in April 2025, the World Health Organization introduced a 6-month all-oral BDLLfxC regimen for multidrug-resistant tuberculosis. Moreover, diarylquinolines, nitroimidazoles, and oxazolidinones such as bedaquiline, pretomanid, and linezolid are under evaluation to enhance efficacy and reduce treatment duration.
The EMR report for the tuberculosis pipeline covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed tuberculosis therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of a few players involved in tuberculosis clinical trials:
This section covers the detailed analysis of each drug under multiple phases, including phase I, phase II, phase III, phase IV, and emerging drugs for tuberculosis. It includes product description, trial ID, study type, drug class, mode of administration, and recruitment status of tuberculosis drug candidates.
JDB0131 Benzenesulfonate is being investigated by WestVac Biopharma Co., Ltd. in a Phase 2b clinical study for drug-resistant tuberculosis. This study is evaluating the efficacy, safety, and pharmacokinetics of oral JDB0131 tablets at varying doses compared with delamanid-based combination regimens. JDB0131 is a novel third-generation nitroimidazole that inhibits mycobacterial cell wall synthesis and generates reactive nitrogen species, enhancing bactericidal activity. The trial is enrolling patients and is expected to reach primary completion by February 2026, with overall study completion anticipated by September 2026.
BNT164a1 and BNT164b1 are investigational mRNA-based tuberculosis vaccines sponsored by BioNTech SE, currently being evaluated in a Phase Ib/IIa study that is examining safety, tolerability, and immunogenicity in BCG-vaccinated, HIV-negative individuals. These vaccines are encoding eight Mycobacterium tuberculosis antigens and are being administered via intramuscular injection in a three-dose schedule. The study is assessing dose escalation and immune response generation, including T-cell activation and antibody production. Researchers identify optimal dosing while monitoring reactogenicity. The primary completion was achieved in March 2026, with overall study completion expected by January 2027.
QTP101 (ID93+GLA-SE) is a recombinant subunit tuberculosis vaccine being developed by Quratis Inc., designed as a multi-antigen protein fusion combining four Mycobacterium tuberculosis antigens with a GLA-SE adjuvant that activates Toll-like receptor 4 to enhance Th1 immune responses. The Phase 1 trial is evaluating safety and immunogenicity in BCG-vaccinated adults aged 55–74 years. The study is administering three intramuscular injections on Days 0, 28, and 56, while monitoring antibody and cytokine responses. The trial is being completed by December 2026, supporting future TB prevention strategies in aging populations.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
The Tuberculosis Pipeline Analysis Report provides a strategic overview of the latest and future landscape of treatments for tuberculosis. It provides necessary information for making informed investment decisions along with research, development, and strategic planning efforts. The stakeholders will benefit from the essential insights into tuberculosis collaborations, regulatory environments, and potential growth opportunities.




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
|
Scope of the Report |
Details |
|
Drug Pipeline by Clinical Trial Phase |
|
|
Route of Administration |
|
|
Drug Classes |
|
|
Leading Sponsors Covered |
|
|
Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Strategic Solutions for Informed Decision-Making
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.