Studying and learning about the functioning process of these microtubules, including control over their own growth, is significant, even though it is a progressive area but still sophisticated, requiring more research work.
The latest study by some researchers from the University of Geneva exhibit that these microtubules could possibly grow and function by techniques that are unfamiliar so far. This study has the potential to contribute to the development of new treatments for diseases that affect our cells. The discoveries from this study were published in the Proceedings of the National Academy of Sciences (PNAS).
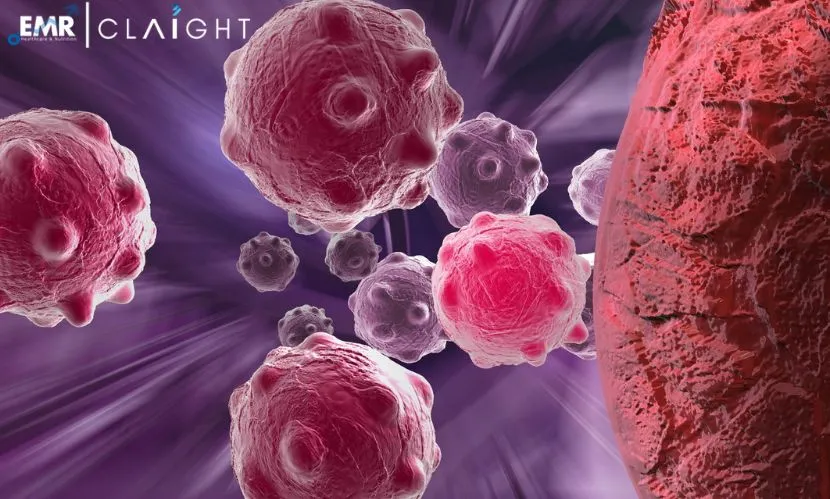
Charlotte Aumeier, the study’s last author, explains, “Microtubules are dynamic structures that continually build and deconstruct themselves. This phenomenon of phase separation, at the microtubule level, increases the concentration of proteins, including tubulin, and significantly stimulates the growth rate of microtubules while reducing depolymerization events, i.e., microtubule decay events.” It is observed that the behaviour of cellular microtubules is directly and physically impacted by this mechanism.
According to the discovery by Julie Miesch, a Ph.D. student, and her team, two proteins, CLIP-170 and EB3, are responsible for controlling the growth of microtubules in cells through a unique liquid phase separation process. It is observed that no communication is encountered between these proteins and microtubules when separated. However, the growth of the microtubule can be controlled by them when coupled with each other. This was observed using an advanced microscopy technique at the UNIGE ACCESS GENEVA platform.
This study has helped several other research enthusiasts to get a better understanding of how things work inside a human cell focusing on microtubules. Additionally, how the teamwork of two proteins CLIP-170 and EB3 contributes massively to controlling the growth of microtubules by separating from the liquid within the cell and forming their own specialized liquid area.
This groundbreaking study has undoubtedly paved the way toward innovative treatments aimed at combating the devastating effects of cancer and degenerative diseases.The key finding of this research involves the identification of a new mechanism governing the growth of microtubules. This mechanism involves two specific proteins, CLIP-170 and EB3, undergoing a process called liquid-liquid phase separation at the tip of the microtubules during their growth. Essentially, these proteins separate from the cellular liquid to form a second liquid phase at the microtubule's tip, like oil droplets in water.This phase separation phenomenon significantly increases the concentration of certain proteins, including tubulin, which in turn, stimulate the growth rate of microtubules, while reducing their breakdown. This newly discovered mechanism appears to play a concrete role in controlling the dynamics of cellular microtubules.The synergy between CLIP-170 and EB3 is crucial for regulating microtubule growth, owing to this phase separation mechanism. CLIP-170 and EB3, when acting together, regulate the speed of microtubule growth at specific locations within the cell.The study used advanced microscopy techniques to observe the role of these two proteins both in vitro and in cellulo, providing insights into their functions.In conclusion, this discovery represents a significant advancement in understanding the regulation of microtubule dynamics within cells. It has the potential to lead to the development of new therapies for conditions like cancer. This breakthrough opens new avenues for research and intervention at the core of cellular processes, promising to deepen our understanding of these crucial mechanisms.
















Share